Translate this page into:
Association of Metformin with Mortality in COVID-19 Patients: A Systematic Review and Meta-Analysis
Address for correspondence Nidhi Agarwal, PhD, Centre for Translational and Clinical Research, School of Chemical & Life Sciences, Jamia Hamdard, New Delhi-110062, India (e-mail: nidhi.bharal@gmail.com).
This article was originally published by Thieme Medical and Scientific Publishers Pvt. Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Studies have demonstrated high prevalence of mortality in coronavirus disease (COVID-19) patients with type 2 diabetes mellitus; however, the effects of antidiabetic pharmacotherapy on COVID-19 complications need further exploration. The aim of the study was to explore the association of metformin use and mortality in COVID-19 patients. A literature search was conducted using the databases Medline (via PubMed) and Cochrane Central Register of Controlled Trials until February 09, 2021. Nine studies were included in the meta-analysis, including 12,684 COVID-19 patients. The meta-analysis suggested 37% lower risk of mortality in patients receiving metformin (risk ratio: 0.63; 95% confidence interval: 0.50–0.78; p < 0.001). However, no significant difference in hospitalization days between the two groups (p = 0.197) was observed. The analysis revealed significantly lower risk of having obesity (p < 0.001), hypertension (p < 0.001), heart failure (p < 0.001), and cerebrovascular disease (p = 0.015) in the group receiving metformin. The analysis also demonstrated significantly lower risk of using anticoagulants (p = 0.015), diuretics (p < 0.001), and antiplatelets (p = 0.010) in patients receiving metformin. Our findings suggest that metformin use decreases mortality in COVID-19 patients. However, randomized studies demonstrating the consequences of metformin use are needed to understand the magnitude of the beneficial effects of metformin.
Keywords
COVID-19
hospitalization
metformin
mortality
meta-analysis
type 2 diabetes
SARS-CoV-2
Introduction
The novel coronavirus disease (COVID-19), now a worldwide public health concern, is associated with varied fatality.1 Some of the recognized risk factors for poor prognosis of COVID-19 patients include age (>65 years), chronic obstructive pulmonary disease (COPD), hypertension, cardiovascular disease, and type 2 diabetes mellitus.2 Although type 2 diabetes mellitus has been reported as a risk factor for COVID-19, the effect of pharmacologic agents used to treat type 2 diabetes mellitus on COVID-19 outcomes remains unclear.
Metformin, the first-line treatment for type 2 diabetes mellitus with adequate safety profile, is being suggested for treatment of COVID-19 as a host-directed therapy.3 A retrospective study highlighted metformin use to be associated with a higher risk of severe COVID-19.3 On the contrary, some retrospective cohort studies have revealed a significant reduction in mortality with metformin use in type 2 diabetes mellitus patients diagnosed with COVID-19.1,4,5,6 The mechanism by which metformin seems to reduce the mortality risk of COVID-19 patients remains unclear. However, its anti-inflammatory actions are hypothesized to be one of the mechanisms.7,8,9
Several studies have demonstrated higher prevalence of mortality in type 2 diabetes mellitus patients with COVID-191,4,5,6; however, the effects of antidiabetic pharmacotherapy on COVID-19 complications need further exploration. Studying the probable effects of pharmacologic agents on clinical prognosis may provide an important understanding to design further studies related to mitigating the risk factors.2 Thus, in this systematic review and meta-analysis, we aimed to explore the association of metformin use and mortality in COVID-19 patients. We hypothesized that mortality would be lower in COVID-19 patients receiving metformin.
Methods
We followed the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) guidelines for systematic reviews10 and Meta-analyses Of Observational Studies in Epidemiology (MOOSE)11 guidelines in designing, conducting, and reporting of this systematic review. The systematic review protocol was registered at PROSPERO (registration ID: CRD42020204384).
Data Sources and Searches
We searched Medline (via PubMed) and Cochrane Central Register of Controlled Trials using the keywords “COVID-19 and metformin” and “2019 novel coronavirus and Metformin.” Additional search was done on LitCovid using the keyword “Metformin” until February 08, 2021. We also searched gray literature using Google Scholar and reference list of eligible articles.
Inclusion and Exclusion
The studies comparing the effect of metformin use on mortality in COVID-19 patients with existing type 2 diabetes mellitus were included. We excluded duplicate publications, reviews, editorials, case reports, letters, meta-analysis, protocols, and studies not reporting the required data. Studies in language other than English were also excluded because of lack of understanding of the data. First author (RP) searched the databases and screened articles for eligibility. Another author (PM) double checked all the included articles and any dispute was resolved by consensus.
Quality Assessment
Two reviewers (RP and RL) assessed the quality of data in the included studies using the National Institutes of Health (NIH) quality assessment tools.12 The NIH tool is comprehensive and widely accepted for assessment of data quality specially in observational study designs. The tools are specific to individual types of included study designs and are designed to assist reviewers in focusing on concepts that are key to critical appraisal of the internal validity of a study. The tools include items for evaluating research question, objective and study population, selection of subjects, sample size justification, exposure, and outcome measures. Quality reviewers could select “yes,” “no,” or “cannot determine/not reported/not applicable” in response to each item on the tool. The overall quality of included studies was rated as good, fair, and poor, and the results were incorporated in the meta-analysis.12
Data Extraction
Data were extracted using a standardized data extraction in Excel and independently checked by a second reviewer (PM) for accuracy. The following variables were extracted: name of the first author, year of publication, study design, age, gender, number of mortalities, comorbidities, and concomitant medications.
Data Synthesis
An exploratory meta-analysis was performed to understand the magnitude and direction of effect estimate. For dichotomous outcomes, risk ratios (RRs) were calculated and presented with respective 95% confidence intervals (CIs). Mantel–Haenszel random-effects meta-analysis using DerSimonian and Laird method was used to pool RRs.13 Continuous outcomes are presented using mean difference (MD) and 95%CIs and pooled using the inverse variance approach. Meta-analysis for each continuous outcome was processed using a random effects model (using DerSimonian and Laird method) since heterogeneity among studies was expected. Heterogeneity between studies was assessed using the χ2-based Cochran's Q statistic (p < 0.1 considered as the presence of heterogeneity) and I-squared (I 2) statistics (>50% representing moderate-high heterogeneity).13 Forest plot was produced, and subgroup analysis was conducted according to the study design. The 95% prediction interval (PI) was calculated, which estimates the uncertainty bounds for a new study evaluating that same association by considering between-study heterogeneity.13 Across the meta-analysis, the statistical significance was set as a p-value of <0.05. Publication bias was not assessed as the total number of included studies in meta-analysis was less than 10.14
Results
Search Results
The systematic search yielded a total of 135 publications using the keyword “Metformin and COVID-19.” After removing duplicates, 86 articles were found to be potential publications for screening. A total of 76 articles (42 review articles and 14 not reporting the required data) were excluded after the application of predefined inclusion and exclusion criteria. Ten studies4,15,16,17,18,19,20,21,22,23 were included for systematic review, while 94,15,16,17,18,20,21,22,23 were included in the meta-analysis (►Fig. 1). All included studies were retrospective cohort studies.
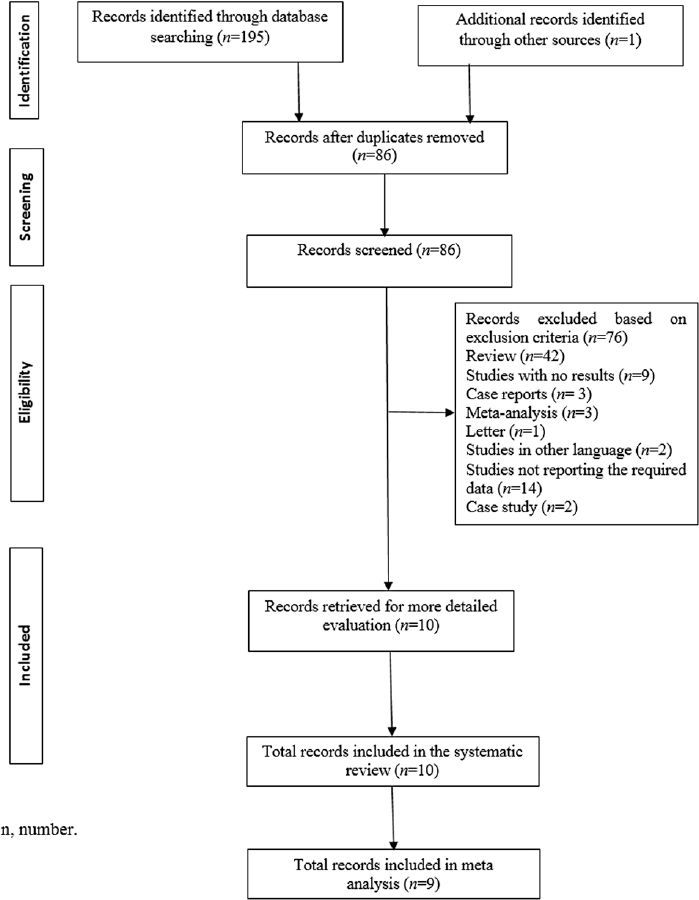
- Prisma flowchart. n, number.
Study Characteristics
All the included studies were retrospective cohort studies.4,15,16,17,18,19,20,22,23 Most of the studies were conducted in China. The studies enrolled a total of 12,684 patients (metformin group: 5,819; no-metformin group: 6,865). The sample size of the studies varied from 120 to 6,250. The study characteristics are provided in ►Table 1.
| Study | Study design | Patient population | Country | Groups | Sample size | Agea | Male, n (%) | Main outcome |
|---|---|---|---|---|---|---|---|---|
| Pérez-Belmonte et al18 | Retrospective cohort | T2DM | Spain | Metformin | 825 | 74.9 (8.4) | 1647 (61.9) | Use of metformin showed no significant association with in-hospital deaths |
| No metformin | 663 | |||||||
| Bramante et al17 | Retrospective cohort | T2DM or obesity | United States | Metformin | 2333 | 73.0 (66.0, 80.0) | 1204 (51.6) | Metformin use not significantly associated with decreased mortality in the overall sample |
| No metformin | 3923 | 76.0 (67.0, 84.0) | 1750 (44.6) | |||||
| Chen, 20204 | Retrospective cohort | T2DM | China | Metformin | 43 | 62.0 (56.0–69.0) | 421 (46.57) | Multivariable regression analyses indicated no association of metformin with in-hospital death |
| No metformin | 77 | 67.0 (57.5–73.0) | ||||||
| Cheng et al19 | Retrospective cohort | T2DM | China | Metformin | 678 | 62.0 (55.0–68.0) | 365 (53.8) | Metformin was not associated with increased mortality |
| No metformin | 535 | 64.0 (58.0–70.0) | 267 (49.9) | |||||
| Crouse et al16 | Retrospective cohort | T2DM | United States | Metformin | 76 | NA | NA | Mortality risk is reduced in subjects taking metformin. Multiple logistic regression analysis revealed metformin to be an independent factor affecting mortality |
| No metformin | 144 | NA | NA | |||||
| Jianget al21 | Retrospective cohort | T2DM | China | Metformin | 100 | 64.0 (56.5–70.0) | 49 (49.0) | No significant association between metformin use and mortality |
| No metformin | 228 | 67.0 (60.0–70.0) | 125 (54.8) | |||||
| Jinghong, 202032 | Retrospective cohort | T2DM | China | Metformin | 37 | 64.6 ± 11.2 | 22 (59.5) | Significant association between metformin use and survival. Multivariate analysis revealed metformin to be an independent predictor of survival |
| No metformin | 94 | 67.7 ± 11.7 | 52 (55.3) | |||||
| Lalauet al22 | Retrospective cohort | T2DM | France | Metformin | 1496 | 68.5 ± 11.9 | 1000 (66.8) | Metformin use is associated with a lower risk of death in patients with diabetes |
| No metformin | 953 | 74.6 ± 12.5 | 568 (59.6) | |||||
| Lally et al23 | Retrospective cohort | T2DM/non-T2DM | United States | Metformin | 127 | 72.3 (8.3) | 125 (98.4) | Subjects taking metformin were at significantly reduced hazard of death |
| No metformin | 69 | 75.6 (9.2) | 68 (98.6) | |||||
| Luo et al31 | Retrospective cohort | T2DM | China | Metformin | 104 | 63.0 (55.8–68.3) | 53 (51.0) | Metformin use was significantly associated with decreased mortality |
| No metformin | 179 | 65.0 (57.5–71.0) | 103 (57.5) |
Abbreviations: N, number of subjects; NA, not available; T2DM, type 2 diabetes mellitus.
a Data presented as mean (standard deviation [SD]) or median (interquartile range [IQR]).
Quality Assessment
We assessed the quality of data in the included studies using the NIH quality assessment tools. The quality assessment indicated that included studies were of acceptable quality. All the papers clearly stated the research question or objective, the study population was clearly specified and defined, and all the subjects were selected from the same or similar populations. The detailed result of the quality assessment is provided in the Supplementary File (available in the online version).
Association between Metformin Use with Mortality and Hospitalization Days
The analysis of the nine retrospective cohort studies suggested that metformin is associated with lower mortality rate (RR: 0.63; 95% CI: 0.50–0.78; p < 0.001; I 2: 73.9%) with substantial heterogeneity in individual study estimates. The results highlight that mortality was 37% lower in patients receiving metformin (►Fig. 2), while Cheng et al19 reported no significant difference in the mortalities of individuals between the metformin and the no-metformin groups (adjusted hazard ratio [HR]: 0.87; 95%CI: 0.36–2.12; p = 0.757). Univariate analysis by Pérez-Belmonte et al18 showed significant association of metformin with mortality (RR: 0.73; CI: 0.58–0.90; p = 0.004). However, the multivariate analysis revealed no significant association (RR: 1.10; CI: 0.76–1.60; p = 0.616). Multiple logistic regression analysis by Crouse et al16 revealed that metformin use is an independent factor affecting mortality in COVID-19 patients. Lally et al23 showed that the patients taking metformin were at significantly reduced hazard of death (adjusted HR: 0.48; 95%CI: 0.28–0.84). Multivariate analysis by Luo et al15 showed that the use of metformin was negatively correlated with the mortality of type 2 diabetes mellitus patients. Four studies qualified for quantitative analysis of hospitalization days. The analysis revealed no significant difference in hospitalization days between the two groups (MD: 1.07; 95%CI: –0.55 to 2.69; p = 0.197; I 2: 0.0; ►Table 2).

- Association of metformin use and mortality in covid-19 patients.
| Outcomes | No. of studies | Sample size | MD | 95% CI | p-value | I-squared (%) | Heterogeneity statistic (p-value) |
|---|---|---|---|---|---|---|---|
| Hospitalization (d) | 4 | 1,646 | 1.07 | −0.55 to 2.69 | 0.197 | 0.0 | 0.18 (0.758) |
| Hematologic parameters (109/L) | |||||||
| White blood cells | 3 | 731 | 0.01 | −0.29 to 0.31 | 0.953 | 0.0 | 1.79 (0.409) |
| Neutrophils | 2 | 403 | −0.11 | −0.48 to 0.26 | 0.561 | 0.0 | 0.61 (0.436) |
| Lymphocytes | 3 | 731 | 0.12 | −0.02 to 0.28 | 0.085 | 39.2 | 3.29 (0.193) |
| Monocytes | 2 | 403 | −0.02 | −0.08 to 0.05 | 0.650 | 51.7 | 2.07 (0.150) |
| Platelets | 2 | 403 | 12.57 | −12.46 to 37.57 | 0.324 | 45.8 | 1.84 (0.174) |
| Eosinophils | 1 | 283 | 0.02 | 0.002 to 0.04 | 0.034 | – | – |
| Biochemical parameters | |||||||
| ALT (U/L) | 3 | 731 | 0.02 | −2.27 to 2.31 | 0.986 | 0.0 | 0.41 (0.815) |
| AST (U/L) | 3 | 731 | −1.66 | −6.11 to 2.78 | 0.464 | 78.3 | 9.20 (0.010) |
| Urea (mmol/L) | 2 | 403 | −0.65 | −1.49 to 0.19 | 0.129 | 53.8 | 2.16 (0.141) |
| LDH (U/L) | 1 | 328 | −12.30 | −28.79 to 4.19 | 0.144 | – | – |
| Ferritin (μg/L) | 1 | 328 | 78.47 | −53.00 to 209.94 | 0.242 | – | – |
| Albumin (g/L) | 1 | 328 | 2.23 | 1.034 to 3.433 | < 0.001 | – | – |
| Triglyceride (mmol/L | 1 | 131 | 0.12 | −0.18 to 0.42 | 0.432 | – | – |
| FBG (mmol/L) | 2 | 611 | 1.00 | −0.59 to 2.58 | 0.218 | 71.7 | 3.53 (0.060) |
| HbA1c (%) | 1 | 328 | 0.70 | 0.27 to 1.13 | 0.001 | – | – |
| HbA1c (mmol/L) | 1 | 131 | 1.77 | 0.98 to 2.56 | < 0.001 | – | – |
| eGFR (mL/min) | 1 | 1,213 | 3.43 | 0.55 to 6.32 | 0.020 | – | – |
| γ-Glutamyltransferase | 1 | 283 | −0.23 | −5.33 to 4.86 | 0.928 | – | – |
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; eGFR, estimated glomerular filtration rate; FBG, fasting blood glucose; HbA1c, hemoglobin A1c; LDH, lactic dehydrogenase; MD, weighted mean difference.
Association between Metformin Use and Laboratory Parameters
The analyzed hematological parameters included white blood cells, neutrophils, lymphocytes, monocytes, platelets, glucose, and eosinophils. The eosinophil level was higher in the group receiving metformin (MD: 0.02; 95%CI: 0.002–0.04; p = 0.034). However, no difference was observed in other hematological parameters (►Table 2). Additionally, higher levels were observed for albumin (MD: 2.23; 95%CI: 1.04–3.43; p < 0.001), HbA1c (%; MD: 0.70; 95%CI: 0.27–1.13; p = 0.001), HbA1c (mmol/L; MD: 1.77; 95%CI: 0.98–2.56; p ≤0.001), and estimated glomerular filtration rate (eGFR; MD: 3.43; 95%CI: 0.55–6.32; p = 0.020) in the group receiving metformin (►Table 2).
Association between Metformin Use with Comorbidities, Clinical Severity, and Oxygen Support
The analysis revealed that the risk of having obesity (RR: 0.53; 95%CI: 0.43–0.65; p < 0.001), hypertension (RR: 0.93; 95%CI: 0.91–0.96; p < 0.001), heart failure (RR: 0.41; 95%CI: 0.33–0.51; p < 0.001), cerebrovascular disease (RR: 0.48; 95%CI: 0.27–0.87; p = 0.015), and COPD (RR: 0.71; 95%CI: 0.62–0.81; p < 0.001) was significantly lower by 48, 7, 59, 53, and 30%, respectively, in the group receiving metformin. No significant difference was observed in clinical severity between the two groups. The risk of using nasal canula was significantly higher in the group not receiving metformin (RR: 1.44; 95%CI: 1.06–1.97; p = 0.02; ►Table 3).
| Outcomes | No. of studies | Sample size | RR | 95% CI | p-value | I-squared (%) | Heterogeneity statistic (p-value) |
|---|---|---|---|---|---|---|---|
| Comorbidities | |||||||
| Overweight | 1 | 28 | 0.56 | 0.24–1.32 | 0.184 | – | – |
| Obese | 1 | 465 | 0.53 | 0.43–0.65 | < 0.001 | – | – |
| Weight loss | 1 | 26 | 0.63 | 0.31–1.29 | 0.210 | – | – |
| Hypertension | 4 | 5,873 | 0.93 | 0.91–0.96 | < 0.001 | 0.0 | 1.85 (0.603) |
| Heart failure | 2 | 282 | 0.41 | 0.33–0.51 | < 0.001 | 0.0 | 0.38 (0.536) |
| Cerebrovascular disease | 1 | 45 | 0.48 | 0.27–0.86 | 0.015 | ||
| Dementia | 2 | 1,071 | 0.81 | 0.58–1.13 | 0.213 | 88.1 | 8.39 (0.004) |
| COPD | 4 | 1,241 | 0.71 | 0.62–0.81 | < 0.001 | 9.3 | 3.31 (0.346) |
| Clinical severity | |||||||
| Moderately ill | 1 | 66 | 1.19 | 0.78–1.83 | 0.421 | – | – |
| Seriously ill | 1 | 207 | 0.98 | 0.84–1.13 | 0.768 | – | – |
| Critically ill | 1 | 10 | 0.43 | 0.09–1.99 | 0.280 | – | – |
| Severe | 1 | 103 | 0.81 | 0.56–1.17 | 0.265 | – | – |
| Nonsevere | 1 | 225 | 1.10 | 0.94–1.27 | 0.237 | – | – |
| Oxygen support | |||||||
| Ambient air | 1 | 66 | 1.19 | 0.78–1.83 | 0.421 | – | – |
| Noninvasive oxygen support | 1 | 211 | 0.97 | 0.84–1.12 | 0.667 | – | – |
| Invasive ventilation | 1 | 9 | 0.62 | 0.13–3.02 | 0.551 | – | – |
| Nasal canula | 1 | 69 | 1.44 | 1.06–1.97 | 0.020 | – | – |
| Nonrebreathing mask | 1 | 7 | 0.17 | 0.01–2.85 | 0.216 | – | – |
| High-flow oxygen | 1 | 7 | 0.42 | 0.05–3.39 | 0.419 | – | – |
| Room air | 1 | 26 | 0.76 | 0.33–1.75 | 0.521 | – | – |
| Noninvasive ventilation | 1 | 19 | 0.68 | 0.24–1.91 | 0.461 | – | – |
| Concomitant medications | |||||||
| Insulin | 4 | 3,427 | 0.85 | 0.59–1.22 | 0.386 | 96 | 75.33 (<0.001) |
| Other oral antidiabetic treatment | 4 | 1,948 | 1.50 | 1.22–1.85 | < 0.001 | 85.8 | 21.06 (<0.001) |
| Chloroquine/hydroxychloroquine | 2 | 80 | 0.75 | 0.31–1.83 | 0.522 | 65.4 | 2.89 (0.089) |
| Anticoagulants | 4 | 2,977 | 0.70 | 0.53–0.94 | 0.015 | 93 | 42.67 (<0.001) |
| Glucocorticoids/steroids | 4 | 1,948 | 0.80 | 0.56–1.16 | 0.240 | 90.7 | 32.14 (<0.001) |
| B-blockers | 2 | 4,268 | 0.82 | 0.61–1.11 | 0.203 | 96.6 | 29.17 (<0.001) |
| Antibacterial treatment | 2 | 1,058 | 1.00 | 0.90–1.10 | 0.975 | 0 | 0 (0.986) |
| Statins | 3 | 5,698 | 1.15 | 1.05–1.27 | 0.004 | 65.4 | 5.79 (0.055) |
| Diuretics | 1 | 989 | 0.69 | 0.63–0.756 | < 0.001 | – | – |
| ARBs and/or ACE inhibitors | 2 | 5,137 | 1.22 | 1.01–1.46 | 0.037 | 95.2 | 20.64 (<0.001) |
| Antiplatelet | 2 | 1,999 | 0.91 | 0.84–0.98 | 0.010 | 0 | 0.26 (0.611) |
| Antivirals | 3 | 796 | 1.08 | 1.03–1.13 | 0.001 | 0 | 1.24 (0.5307 |
| Overall | 33 | 30,325 | 0.95 | 0.86–1.04 | 0.276 | 96.8 | 1,008.44 (<0.001) |
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blockers; CI, confidence interval; COPD, chronic obstructive pulmonary disease; RR, relative risk.
Association between Metformin Use and Concomitant Treatments
The analysis revealed that the risk of using anticoagulants (RR: 0.70; CI: 0.53–0.94; p = 0.015), diuretics (RR: 0.69; C95%CI: 0.63–0.76; p < 0.001), and antiplatelets (RR: 0.91; 95%CI: 0.84–0.98; p = 0.010) was significantly lower by 30, 32, and 10%, respectively, in patients receiving metformin (►Table 3).
Discussion
Emerging evidence suggest that the presence of comorbidities increases the mortality risk in COVID-19 patients. Diabetes forms an important risk factor to predict the adverse outcomes.1 Several studies have demonstrated high mortality rate in type 2 diabetes mellitus patients1,5,6,24; however, the effect of antidiabetics on the mortality remains to be elucidated.25 Thus, the present meta-analysis was conducted to assess the association of metformin use with mortality in COVID-19 patients. The present meta-analysis was based on data from nine studies on COVID-19 patients. The analysis demonstrated lower mortality in the group receiving metformin; however, no significant difference in hospitalization days was observed between the two groups. The analysis also revealed significant difference in the prevalence of obesity, hypertension, heart failure, cerebrovascular disease and COPD. No significant difference was observed in clinical severity between the two groups.
Several retrospective cohort studies have demonstrated low mortality in patients receiving metformin.15,17,24,25 In line with our findings, other meta-analysis have revealed a significantly lower mortality in patients receiving metformin.7,26 Retrospective cohort studies have shown metformin use to be an independent factor associated with mortality.16,20 However, retrospective cohort studies have revealed no difference in hospitalization days.20,21 Additionally, no difference in the clinical severity between the groups receiving and not receiving metformin was revealed in retrospective cohort studies.15,21 Furthermore, retrospective cohort studies have reported a lower number of patients with obesity,17 hypertension,17,22 heart failure,19,22 cerebrovascular disease,19 and COPD22 in patients receiving metformin.
The potential mechanisms underlying the reduction of mortality by metformin in COVID-19 patients remain unclear.7 However, several mechanisms have been proposed for the same. Metformin is thought to increase the expression of angiotensin converting enzyme 2 (ACE2),2,27,28,29 a receptor for severe acute respiratory syndrome coronavirus 2.2 Metformin has been demonstrated to activate adenosine monophosphate-activated protein kinase (AMPK) in in vitro and in vivo experiments,2,7 thus regulating ACE2 and protein stability.2 Activation of AMPK further leads to the inhibition of nuclear factor kappa B (NF-kB), which is a transcription regulator involved in inflammation. Inhibition of NF-kB subsequently inhibits the production of proinflammatory cytokines, such as interleukins 8 and 1α.7 Metformin is also suggested to have antithrombotic effects. Evidence suggests excessive inflammatory responses as well as disseminated thromboembolic events are associated with increased mortality in COVID-19 patients.30 Therefore, metformin is suggested to have beneficial effect by its antifibrinolytic activities30 and inhibiting the cytokine storm in COVID-19 patients.7 Additionally, metformin induces autophagy, thus controlling inflammation and activating innate and adaptive immunity.31 Furthermore, metformin is suggested to act on Na+/H+ exchangers and vacuolar ATPase, subsequently inhibiting viral infection by raising the cellular pH leading to interference with the endocytic cycle.
This study had several limitations. A small number of studies have been included in the meta-analysis; retrospective cohort studies were included, and substantial heterogeneity was observed among the studies, which poses a significant limitation for the conclusions drawn. Although an extensive search was done, studies might have been missed inadvertently. Exclusion of studies in languages other than English might have resulted in missing of relevant studies.
Conclusion
To conclude, our findings suggest that metformin may contribute to reduced mortality in COVID-19 patients. However, randomized studies demonstrating the effects of metformin in COVID-19 are needed to support clinical recommendations of metformin in high-risk population. Further investigations are warranted to look into the likelihood of metformin improving prognosis of COVID-19.
Authors Contribution
All the authors contributed in conception or design. RP, PM, and RL contributed in acquisition of data. RB and NA contributed in data analysis and interpretation. All the authors contributed to the preparation of manuscript or critically revising the manuscript and gave their final approval for the manuscript.
Conflict of Interest
None declared.
Funding
None.
References
- Association of diabetes and hypertension with disease severity in covid-19 patients: a systematic literature review and exploratory meta-analysis. Diabetes Res Clin Pract. 2020;166:108295.
- [CrossRef] [PubMed] [Google Scholar]
- Is metformin a miracle or a menace in COVID-19 patients with type 2 diabetes? J Diabetes Investig. 2021;12(04):479-481.
- [CrossRef] [PubMed] [Google Scholar]
- Risk of metformin in patients with type 2 diabetes with COVID-19: a preliminary retrospective report. Clin Transl Sci. 2020;13(06):1055-1059.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368:m1091.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062.
- [CrossRef] [PubMed] [Google Scholar]
- CORONADO investigators. Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study. Diabetologia. 2020;63(08):1500-1515.
- [CrossRef] [PubMed] [Google Scholar]
- Mortality risk with preadmission metformin use in patients with COVID-19 and diabetes: a meta-analysis. J Med Virol. 2021;93(02):695-697.
- [CrossRef] [PubMed] [Google Scholar]
- Activation of the AMP-activated kinase by antidiabetes drug metformin stimulates nitric oxide synthesis in vivo by promoting the association of heat shock protein 90 and endothelial nitric oxide synthase. Diabetes. 2006;55(02):496-505.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin inhibits cytokine-induced nuclear factor kappaB activation via AMP-activated protein kinase activation in vascular endothelial cells. Hypertension. 2006;47(06):1183-1188.
- [CrossRef] [PubMed] [Google Scholar]
- PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(07):e1000097.
- [Google Scholar]
- Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008-2012.
- [CrossRef] [PubMed] [Google Scholar]
- Study Quality Assessment Tools. National Heart, Lung and Blood Institute n.d at: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed )
- [Google Scholar]
- Publication bias: the elephant in the review. Anesth Analg. 2016;123(04):812-813.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis. Am J Trop Med Hyg. 2020;103(01):69-72.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin use is associated with reduced mortality in a diverse population with Covid-19 and diabetes. Front Endocrinol (Lausanne). 2021;13(11):600439.
- [CrossRef] [PubMed] [Google Scholar]
- Observational study of metformin and risk of mortality in patients hospitalized with Covid-19. Lancet Healthy Longev. 2021;2(01):e34-e41.
- [CrossRef] [PubMed] [Google Scholar]
- SEMI-COVID-19 Network. Mortality and other adverse outcomes in patients with type 2 diabetes mellitus admitted for COVID-19 in association with glucose-lowering drugs: a nationwide cohort study. BMC Med. 2020;18(01):359.
- [Google Scholar]
- Metformin is associated with higher incidence of acidosis, but not mortality, in individuals with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;32(04):537-547.e3.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin use in diabetes prior to hospitalization: effects on mortality in Covid-19. Endocr Pract. 2020;26(10):1166-1172.
- [CrossRef] [PubMed] [Google Scholar]
- Association of metformin with mortality or ARDS in patients with COVID-19 and type 2 diabetes: a retrospective cohort study. Diabetes Res Clin Pract. 2021;173:108619.
- [CrossRef] [PubMed] [Google Scholar]
- CORONADO investigators. Metformin use is associated with a reduced risk of mortality in patients with diabetes hospitalised for COVID-19. Diabetes Metab. 2021;47(05):101216.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin is associated with decreased 30-day mortality among nursing home residents infected with SARS-CoV2. J Am Med Dir Assoc. 2021;22(01):193-198.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical Characteristics and outcomes of patients with diabetes and COVID-19 in association with glucose-lowering medication. Diabetes Care. 2020;43(07):1399-1407.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin use in relation to clinical outcomes and hyperinflammatory syndrome among COVID-19 patients with type 2 diabetes: a propensity score analysis of a territory-wide cohort. Front Endocrinol (Lausanne). 2022;13:810914.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin use is associated with reduced mortality rate from coronavirus disease 2019 (COVID-19) infection. Obes Med. 2020;19:100290.
- [CrossRef] [PubMed] [Google Scholar]
- The angiotensin-converting enzyme 2 (ACE2) receptor in the prevention and treatment of COVID-19 are distinctly different paradigms. Clin Hypertens. 2020;26:14.
- [CrossRef] [PubMed] [Google Scholar]
- Diabetes, metformin and the clinical course of Covid-19: outcomes, mechanisms and suggestions on the therapeutic use of metformin. Front Pharmacol. 2022;13:784459.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin in COVID-19: clinical trials are needed to prove its benefits. Ir J Med Sci. 2022;191(06):2641-2642.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin use is associated with reduced mortality in a diverse population with COVID-19 and diabetes. Front Endocrinol (Lausanne). 2021;11:600439.
- [CrossRef] [PubMed] [Google Scholar]
- A proposed mechanism for the possible therapeutic potential of Metformin in COVID-19. Diabetes Res Clin Pract. 2020;167:108282.
- [CrossRef] [PubMed] [Google Scholar]
- Metformin Use in Diabetes Prior to Hospitalization: Effects on Mortality in Covid-19. Endocr Pract. 2020;26(10):1166-1172.
- [Google Scholar]






