Translate this page into:
Safety and efficacy of TightRail mechanical sheath for transvenous lead extraction
* Corresponding author: Dr. Vijay Bohra, Department of Cardiology, Command Hospital, Central Command, Lucknow Cantt, Lucknow, Uttar Pradesh, India. aadivijay@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Bohra V, Aggarwal N, Keshavamurthy G, Guleria V, Singh B. Safety and efficacy of TightRail mechanical sheath for transvenous lead extraction. Ann Natl Acad Med Sci (India). 2025;61:127-31. doi: 10.25259/ANAMS_22_2024
Abstract
Objectives
This study aimed to evaluate the safety and efficacy of the bidirectional rotational mechanical sheath TightRail (Spectranetics Corp., Colorado Springs, CO, USA) for lead extraction in an Indian center.
Material and Methods
A prospective study of patients who underwent transvenous lead extractions (TLE) in whom manual extraction was ineffective during the period from March 2018 to March 2021.
Results
A total of 28 patients underwent lead extraction using TightRail. The mean age at the time of extraction was 62.4±18.07 years. The average duration between implantation and extraction was 8.8±4.6 years. The most common etiology was pocket site infection (78.6%), followed by lead endocarditis (7.1%), chronic pain (3.6%), and lead fracture (10.7%). In total, 53 lead extractions were attempted, with 51 procedures, resulting in a clinical success rate of 96.2%. Approximately 7.14% patients required blood transfusions, and 3.6% developed pericardial effusion. There was no incidents of cardiac avulsion, death or arrhythmias requiring cardioversion.
Conclusion
This extensive clinical study in India demonstrated that the use of TightRail provides high safety and efficacy for lead extractions across a wide range of lead age. The incidence of infectious etiology was found to be much higher in Indian patients compared to Western literature.
Keywords
Lead extraction
Pacemaker
TightRail
INTRODUCTION
In recent years there has been a surge in the use of cardiac implantation electronic devices (CIED). Concurrently, infections and lead malfunctions associated with these devices have led to a rise in transvenous lead extractions (TLE).1 Despite advancements in technologies and techniques, TLE continues to be a challenging procedure, carrying the risk of potentially life-threatening complications.2 Chronically implanted leads often develop fibrotic adhesions, which further complicate the TLE process. Byrd et al. (1999) reported that the likelihood of failure or partial removal doubles for every 3 years of implant duration.3 Numerous studies have demonstrated the safety, efficiency, and high success rates of mechanical rotational TLE devices in extracting chronically implanted leads.4,5 TightRail (Spectranetics Corp., Colorado Springs, CO, USA) is a bidirectional rotational mechanical sheath with a flexible shaft that ensures high coaxiality with the lead and a dilating blade that remains shielded until activated. However, there is a limited number of studies from India documenting the safety and effectiveness of mechanical TLE. This study aims to share our experience with the TightRail sheath for lead extractions in India.
MATERIAL AND METHODS
Study design and procedure
This prospective study included all patients who underwent TLE in whom manual traction was ineffective, and where the TightRail bidirectional rotational mechanical sheath was utilized, during the period from March-2018 to March 2021 in India.
A subclavian approach was adopted for all patients; if a patient was pacemaker (PM) dependent, a temporary PM was inserted via the femoral vein. The leads were carefully dissected from adhesions within the generator pocket and the fixation sutures were removed [Figure 1a]. Initially, simple traction was attempted in all cases. If this was unsuccessful, a locking stylet (Spectranics Corp., LLDTM lead locking device) was introduced into the inner lumen of the lead and deployed [Figure 1b]. If traction using the locking stylet did not work, the TightRail dilator sheath was then used [Figure 2]. Only cases that required this rotational dilator sheath were included in the study. The TightRail dilator sheath is composed of outer and inner shafts, with the inner shaft capable of rotating within the stationary outer shaft via a handled mechanism. The tip of the inner shaft has blades designed to cut and dilate the fibrous attachments to the leads, facilitating extraction [Figure 3].
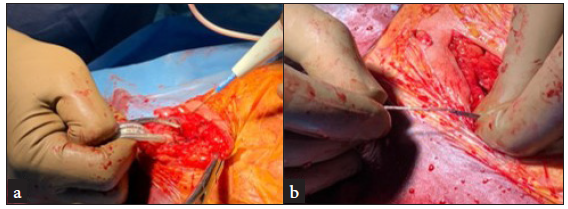
- (a) Leads being dissected free from adhesions inside the generator pocket, (b) A lead locking stylet being introduced into the inner lumen of lead.
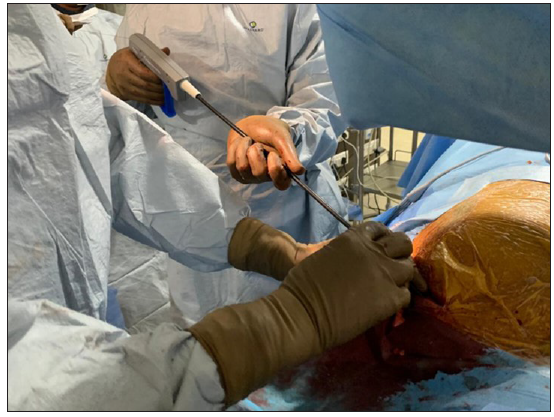
- TightRail dilator sheath in use.
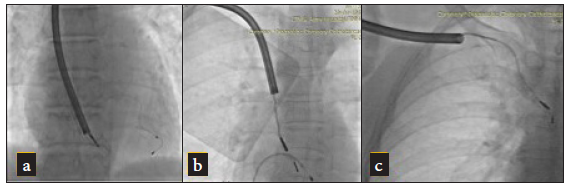
- (a–c) Angiogram showing TightRail dilator sheath being used to extract a right ventricular lead (a) distal end of the lead, (b) SVC & RA junction, (c) at right subclavian vein.
Study endpoints
The primary endpoints of this study were to assess the rates of clinical success and complete procedural success. Clinical success was defined as the removal of all targeted leads and lead material from the vascular space, or retention of a small portion of the lead (fragment <4 cm) that did not impact the overall outcome of the procedure. Complete procedural success was defined as the removal of the targeted lead and all lead material from the vascular space, accompanied by the absence of permanently disabling complications or procedure-related mortality.6 All procedure-related complications were noted.
Statistical analysis
Categorical variables are presented as frequencies and percentages, while continuous variables are presented as mean ± standard deviation (SD).
RESULTS
During the study period, a total of 28 patients underwent lead extraction using the TightRail bidirectional rotational mechanical extraction sheath. As shown in Table 1, there was a male preponderance, with 18 males (74%) and 10 females (36%) in the study population. The mean age at the time of extraction was 62.4±18.07 years. The average duration from the time of implantation to extraction was 8.8±4.6 years, with a maximum of 13.5 years and a minimum of 5 years. The indications for lead extraction in the study are presented in Table 2. Among the various etiologies, pocket site infection was the most common etiology for CIED extraction, accounting for 22 patients (78.6%).
| Variables | N = 28 patients |
|---|---|
| Age (Mean ± SD, years) | 62.4±18.07 |
| Males, n (%) | 18 (64%) |
| Females, n (%) | 10 (36%) |
| Diabetes mellitus, n (%) | 17 (60%) |
| Coronary artery disease, n (%) | 12 (45%) |
| Chronic obstructive pulmonary disease, n (%) | 6 (20%) |
| Ejection fraction (Mean ± SD, %) | 35 ± 16.3 |
SD: Standard deviation, N: Total number of patient in the study, n: Number of patients in the subgroups
| Etiology | N=28 patients |
|---|---|
| Pocket site infection, n (%) | 22 (78.6%) |
| Lead endocarditis, n (%) | 2 (7.1%) |
| Chronic pain, n (%) | 1 (3.6%) |
| Lead fracture, n (%) | 3 (10.7%) |
Among the explanted CIED [Table 3], Dual chamber PM (DDDR) devices were the most common, accounting for 35.7% (10 patients). In total 53 lead extractions were attempted in this study, with 79.25% of the leads being of the screwing type. Of these attempts, 51 leads were successfully extracted, achieving a complete procedural success rate of 94.33% and a clinical success rate of 96.22%.
| Type of CIED | Number of patients (%) | Pocket site infection | Lead endocarditis | Chronic pain | Lead fracture |
|---|---|---|---|---|---|
| DDDR | 10 (35.7%) | 8 | 1 | 1 | - |
| AICD-D | 7 (25%) | 5 | 1 | - | 1 |
| VVIR | 6 (21.5%) | 5 | - | - | 1 |
| CRT-D | 2 (7.14%) | 2 | - | - | - |
| CRT-P | 1 (3.6%) | 1 | - | - | - |
| AICD-S | 2 (7.14%) | 1 | 1 |
AICD: Automated implantable cardioverter defibrillator, CIED: Cardiac implantable electronic devices, CRT-D: Cardiac resynchronization therapy – defibrillator, CRT-P: Cardiac resynchronization therapy – pacemaker, DDDR: Dual chamber pacemaker, VVIR: Single chamber pacemaker, S: Single chamber, D: Dual chamber.
As shown in Table 4, the study included 20 right atrial, 28 right ventricular, and 3 left ventricular lead extractions. One patient underwent five lead extractions related to a cardiac resynchronization therapy-defibrillator (CRT-D) upgrade from a dual-chamber automated implantable cardiac defibrillator (including 2 right ventricle, 2 right atrial, and 1 left ventricular). However, one right atrial lead from a DDDR PM implanted 11 years ago and a right ventricular lead from a V paced V sensed single chamber PM (VVVIR) PM implanted 12 years ago could not be extracted. Both the unextracted leads were of screwing type. Moreover, one patient had a cardiac resynchronization therapy-PM (CRT-P) right ventricular lead (screwing, less than 4 cm) and a left ventricular lead (more than 4 cm) that were partially extracted [Figure 4].
| Type of CIED | Number of devices | Right atrial leads extracted | Right ventricular leads extracted | Left ventricular leads extracted | Total leads extracted | Leads couldn’t be extracted | Leads partially extracted |
|---|---|---|---|---|---|---|---|
| DDDR | 10 | 9 | 10 | - | 19 | 1 | - |
| AICD-D | 7 | 7 | 7 | - | 14 | - | - |
| VVIR | 6 | - | 5 | - | 5 | 1 | - |
| CRT-D | 2 | 3 | 3 | 2 | 8 | - | - |
| CRT-P | 1 | 1 | 1 | 1 | 3 | - | 1 |
| AICD-S | 2 | - | 2 | - | 2 | - | - |
| TOTAL | 28 | 20 | 28 | 3 | 51 | 2 | 1 |
AICD: Automated implantable cardioverter defibrillator, CIED: Cardiac implantable electronic devices, CRT-D: Cardiac resynchronization therapy – defibrillator, CRT-P: Cardiac resynchronization therapy – pacemaker, DDDR: Dual chamber pacemaker, VVIR: Single chamber pacemaker, S: Single chamber, D: Dual chamber.
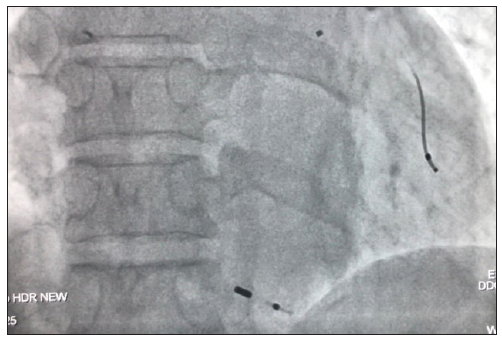
- Partially extracted left and right ventricular leads.
Two patients in this study required blood transfusion, and one patient developed moderate pericardial effusion, which was managed conservatively. There was no cases of cardiac avulsion, mortality, or arrhythmias requiring cardioversion [Table 5].
| Events | N (%) |
|---|---|
| Death | - |
| Bleeding requiring transfusion | 2(7.14%) |
| Hematoma requiring drainage | - |
| Cardiac avulsion or tear requiring surgery | - |
| Pericardial effusion | 1(3.6%) |
| Arrhythmias requiring cardioversion | - |
DISCUSSION
The term “lead extraction” refers to the use of specialized equipment to extract a lead that is at least one year old. In contrast, the term “lead explantation” is used for leads that are less than one year old and do not require special equipment.2 The first documented case of lead extraction was reported in 1968. The procedure is complex, but the development of locking stylets has improved procedural success rates. The introduction of laser-assisted TLE devices has further improved these success rates.7,8 These laser system utilize excimer lasers to break tissue bonds and vaporize the fibrotic bands surrounding the target leads. However, laser-assisted TLEs generally demonstrate lower clinical and procedural success rates compared to mechanical rotational sheaths.9 Mechanical rotational sheaths, introduced in 2006, employ blades that effectively free chronically implanted leads. Numerous studies have shown that mechanical rotational TLE devices are efficient and have high success rates,4,6 as well as reduced mortality risk compared to laser sheaths.
TLE has allowed us to achieve successful and safe extraction of leads in patients across a wide range of ages. The demographic profile of our study participants, including a mean age of 62.4 years and a preponderance of male patients, reflects a high prevalence of, diabetes mellitus (60%), and coronary artery disease (45%), which is consistent with findings from other TLE studies.10
The mean duration of lead implantation in our study was 8.8 ±4.6 years. Notably, pocket site infection and lead endocarditis constituted 85.7% of the extraction cases, which is much higher compared to other studies. In the 2010 LExICon study, infectious indications were reported in 56.9% of the extracted leads.8 Similarly, the authors of the ELECTRa study11 (2017) and a large, multicentre Italian registry4 (2018) reported infectious indications in 52.8% and 50.8% of lead extractions, respectively.
We achieved a complete procedural success rate of 94.33% and a clinical success rate of 96.22%. Compared with similar TLE studies, our research included patients with longer lead indwell times and reports better success rates. The rate of procedure-related major complications in the LExICon study was reported to be 0.9%.8 Our study did not experience any major complications. Minor complications were observed in 10.74% patients, with 7.14% requiring blood transfusion due to localized bleeding and 3.6% experiencing moderate pericardial effusion. Data from the National Cardiovascular Data Registry, which included 11,304 ICD extractions, revealed that only 0.36% of patients required urgent cardiac surgery; however, these emergent procedures had a 34% mortality rate.12 Our study is among the few in the literature documenting the use of Tightrail in Indian patients. Sawhney et al. (2016) reported successful TLE using TightRail in three patients.13
CONCLUSION
This study, the largest clinical investigation of Indian patients to date, demonstrates that the use of TightRail provides high safety and efficacy for lead extractions across a wide range of lead age. Infectious etiologies were found to be significantly more prevalent in Indian patients compared to Western literature.
Authors’ contributions
VB: Conception, design, materials & method, analysis, writing literature review; NA: Critical review; GK: Supervision; VG: Material & method, analysis, writing, literature review; BS: Literature review.
Ethical approval
The research/study approved by the Institutional Review Board at Army Hospital (Research & Referral), New Delhi, number IEC-18/17, dated 23rd December 2017.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Trends in permanent pacemaker implantation in the United States from 1993 to 2009: Increasing complexity of patients and procedures. J Am Coll Cardiol. 2012;60:1540-5.
- [CrossRef] [PubMed] [Google Scholar]
- 2017 HRS expert consensus statement on cardiovascular implantable electronic device lead management and extraction. Heart Rhythm. 2017;14:e503-51.
- [CrossRef] [PubMed] [Google Scholar]
- Intravascular extraction of problematic or infected permanent pacemaker leads: 1994-1996. U.S. Extraction Database, MED Institute. Pacing Clin Electrophysiol. 1999;22:1348-57.
- [CrossRef] [PubMed] [Google Scholar]
- Safety and efficacy of the new bidirectional rotational evolution® mechanical lead extraction sheath: Results from a multicentre Italian registry. Europace. 2018;20:829-34.
- [CrossRef] [PubMed] [Google Scholar]
- Lead extractions: The Zwolle experience with the evolution mechanical sheath. Europace. 2016;18:762-6.
- [CrossRef] [PubMed] [Google Scholar]
- Success and complication rates of lead extraction with the first- vs. the second-generation Evolution mechanical sheath. Europace. 2017;19:1717-22.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical study of the laser sheath for lead extraction: The total experience in the United States. Pacing Clin Electrophysiol. 2002;25:804-8.
- [CrossRef] [PubMed] [Google Scholar]
- Lead extraction in the contemporary setting: The LExICon study: an observational retrospective study of consecutive laser lead extractions. J Am Coll Cardiol. 2010;55:579-86.
- [CrossRef] [PubMed] [Google Scholar]
- Advanced techniques for chronic lead extraction: Heading from the laser towards the evolution system. Europace. 2013;15:1771-6.
- [CrossRef] [PubMed] [Google Scholar]
- Results of the Patient-Related Outcomes of Mechanical lead Extraction Techniques (PROMET) study: A multicentre retrospective study on advanced mechanical lead extraction techniques. Europace. 2020;22:1103-10.
- [CrossRef] [PubMed] [Google Scholar]
- The European Lead Extraction ConTRolled (ELECTRa) study: A European Heart Rhythm Association (EHRA) registry of transvenous lead extraction outcomes. Eur Heart J. 2017;38:2995-3005.
- [CrossRef] [PubMed] [Google Scholar]
- The role of transvenous lead extraction in the management of redundant or malfunctioning pacemaker and defibrillator leads post ELECTRa. Europace. 2018;20:1733-40.
- [CrossRef] [PubMed] [Google Scholar]
- Percutaneous lead extraction and venous recanalisation using spectranetics tight rail: A single centre experience. Indian Pacing Electrophysiol J. 2016;16:134-8.
- [CrossRef] [Google Scholar]






