Translate this page into:
Current Concepts of Pathophysiology of Vasovagal Syncope and its Evaluation and Management: A Review
Former Senior Adviser (Aero Space Medicine) and Professor of Physiology, Institute of Aerospace Medicine IAF, Bunguluru-560017 and Former Professor & Head, Department of Physiology, College of Medicine and SQUH Hospital, Sultan Qaboos University, Muscat, Oman.
Correspondence: Gp Capt (Rtd) Prof. Mohan B Dikshit, 48 Shivaji Housing Society, Senapati Bapat Road, Pune - 411016, India. Email: justmohan@gmail.com
Abstract
Vasovagal syncope (VVS) with a sudden, temporary loss of consciousness (LoC) is a common phenomenon in the young and the elderly. Though generally described as innocuous, it may lead to serious consequences in special category of people (pilots), or in the elderly in whom LoC may lead to a fall and serious injury. The topic has been copiously researched upon and discussed in medical literature over the last few decades, but the exact mechanisms which lead to the disability have yet to be fully agreed upon. Changes in cardiovascular baroreceptor sensitivity, aberrations in the complex interaction amongst the nucleus of the tractus solitarius and the nuclei around it, inability of the peripheral circulation to respond to autonomic vasoconstrictors, or excess production of vasodilators such as nitric oxide produced locally have been considered in its pathophysiology. Various extraneous situations like dehydration, exposure to heat stress, medications, psychological factors may adversely stress regulatory physiological responses and promote occasional episodes of VVS. More complex dysautonomia could be a reason for the recurrent VVS. Differences between brain structure of VVS sufferers and normal subjects have been proposed. Head-up tilt table (HUT) test is the most widely applied investigation for evaluating VVS episodes. Lower body negative pressure (LBNP) has also been used. Enhancement of the orthostatic stress may be done by simultaneous use of both, or with peripheral vasodilators. As to whether such an enhancement is necessary is debatable. Management with increased salt and fluid intake, corticosteroids, beta adrenergic receptor blockers, alpha adrenergic receptor stimulants, selective serotonin reuptake inhibitors, and nitric oxide synthase inhibitors have been tried with variable success.
Keywords
Vasovagal syncope
head-up tilt table
loss of consciousness
pilot
dysautonomia
orthostatic stress.
Introduction
Vasovagal syncope (VVS), a sudden loss of consciousness in apparently healthy individuals, though brief, and medically innocuous, is often interpreted as a catastrophic event by the sufferer. Recovery is full and rapid. It is more a symptom complex than a disease, but may leave behind a lurking doubt in the individual's mind about his/her sense of well being. There may be a loss of confidence in one's over all ability which becomes particularly relevant in certain categories of people such as aviators who may fear for their professional careers, further escalating the situational anxiety. Undeniably if a loss of consciousness occurs because of causes listed as “pathological” in Table 1, the matter is of grave concern. However, what is addressed in this review is the VVS which is a temporary aberration of body physiology, the mechanism of which is still as elusive as the Scarlet Pimpernel. Although the loss of consciousness (LoC) during VVS is “brief” and recovery is “spontaneous and complete”; in an occupation such as aviation, the condition may have life threatening consequences (1).
| Pathological |
| 1. Cardiovascular disease producing a sudden fall in cardiac output |
| 2. CNS disorders such as transient ischemic attacks |
| 3. Autonomic dysfunction: diabetes mellitus, postural tachycardia syndrome (POTS), shy-dragers syndrome |
| Physiological |
| 1. Postural change from supine to upright |
| 2. Prolonged standing |
| 3. Heat stress |
| 4. Dehydration/hemorrhage |
| 5. Pain |
| 6. Anxiety/fear |
| 7. Micturition syncope |
| 8. Medications |
| 9. Insomnia/fatigue |
| 10. Age related |
| 11. Post space flight |
| 12. Pregnancy |
| Most of the above (listed as Physiological) may produce a rapid reduction in the central blood volume leading to a sudden reflex decrease in heart rate in combination with peripheral. |
Definition and Types of Syncope
Syncope has been variously defined as “a temporary loss of consciousness” (2); “A sudden transient loss of consciousness with an inability to maintain postural tone that was not compatible with a seizure disorder, vertigo, coma, shock, and other states of altered consciousness” (3); “A sudden and brief loss of consciousness and postural tone from which recovery is spontaneous (4), or more recently as “A temporary loss of consciousness and postural tone” (5) or as “A sudden and transient loss of consciousness with spontaneous recovery” (6). It is obvious that basic concept of what defines syncope has remained unchanged over a period of about six decades, and is unlikely to do so in the future. Collectively stated: “Syncope is a short duration, reversible loss of consciousness and postural tone following a rapid fall in blood pressure from which an individual recovers spontaneously, usually without any residual effects. In most instances it is associated with a change of posture from supine to erect”.
Incidence
Vasovagal syncope, or the “common faint” occurs in apparently healthy young people (more in females, and during adolescence) with an incidence which has varied from about 6% (7) to as high as 25% (8). A very extensive report by da Silva (2014) (9) cites an incidence which varies from about 15% in children, to 39% in young adults and 23% in the elderly. Syncopes are classified as reflex (vasovagal), cardiac, neurological and others, with the vasovagal syncope as the most frequent. The percentage of VVS has not been clarified in the overall percentages quoted in this study. Lavania et al (10) using a questionnaire survey found a fairly high incidence of presyncope/ syncope (about 30%) in 134 healthy medical students of both sexes, mean age 19.3 years. However, their findings were confounded by the fact that none of the 41 students who had given positive history of a faint developed even presyncopal symptoms when subjected to passive upright standing. In our series of 166 head-up tilt table (HUT) exposures of 143 healthy military men (mean age 29.4 years) without a prior history of orthostatic intolerance, there was not a single episode of presyncope/ syncope. However, 22.8% of the 57 young, apparently healthy airmen with previous history of syncope/presyncope referred to our laboratory for evaluation, developed some symptoms of orthostatic intolerance, though not frank syncope during HUT (11). It is thus obvious that VVS is indeed an enigma as suggested by Dikshit (1) and its mode of occurrence remains far from being explained suitably. In apparently healthy elderly population (6th to 8th decades of life) a 6% to 11% incidence of unexplained syncope has been reported (6). Incidence in children may be as high as 10%-44% (12). Information on syncope in Indian children, and to that matter on cardiovascular response to orthostatic stress in normal Indian children is for all practical purposes unavailable.
Mammals other than man are generally not known to develop VVS as they do not need to operate in an upright posture as humans do. And even if they do so (giraffes reaching out for food by raising their necks to significant heights), the relatively low blood flow to their proportionately small brains size makes them more or less immune to below par perfusion of their brains (13). There is no known report of orthostatically induced VVS in the great apes, (our ancient cousins) probably again because of the relatively small size of their brain, and also the fact that they prefer to operate on all fours as a routine rather than in the erect posture which they may assume at will. Thus, it is almost impossible to use an animal model to study the intricacies of the physiology of postural change induced syncope. Emotional or fear-induced bradycardia and hemorrhage/severe fluid loss, though known to produce conditions close to the VVS seen in humans, but a loss of consciousness is not known to occur (14).
Gender and Race Differences in Occurrence of Syncope
Little information is available on these issues. Almost all our data on response to orthostatic stress and incidence of syncope has been obtained in male subjects (1, 11, 15-23). Patel et al (24) were unable to demonstrate gender difference in their male and female subjects during exposure to HUT. They did not report any incidence of syncope. On the other hand, Sachse (25) reported a higher incidence of orthostatic intolerance during 6 minutes of standing after supine rest in elderly women as compared to males in the same age group. Frank syncope was not seen. It is difficult to draw any major conclusions from her relatively limited study as she also reported that heart rate variability in both her groups (males and females) was similar. A large Korean study (26) comprising of 497 males and 547 females reported that females had a higher incidence of syncope/presyncope. A high incidence in young women has also been reported by others (6). da Silva (9) on the other hand while reviewing a very large data, opined that there was no gender difference. She had concentrated more on the overall incidence of various types of syncope conceding that the VVS was the most common variety though specific incidence for this entity was not recorded.
Little information is available on racial differences in the occurrence of syncope. Goldstein and Shapiro (27) reported that Blacks have a different response to orthostatic stress as compared with Asians and Whites, but there was no mention of VVS in their study. Some of our own observations are tabulated in Table 2. Some minor differences in the normal responses of the recorded parameters were seen though statistical comparisons were not made. Our orthostatic tolerance tests using either HUT (11, 20-23) or lower body negative pressure (LBNP) (19) were performed on normal healthy, physically fit military men. None of the subjects exposed to the stress for the conventional period of 20 minutes (HUT) or a maximum LBNP-50 mmHg for 5 minutes, showed any symptoms of discomfort or signs of impending VVS. However, as stated earlier 22.8% of the 57 military men who were evaluated in our laboratory for single episodes of presyncope /syncope did show abnormal response to HUT (11). A number of questions arise out of these findings:
Can these individuals be considered as a part of the so called “normal” population or condemned outright as those with abnormal cardiovascular reflex status?
Were they victims of a one off aberration in response to any one or more of the situational stresses listed under “Physiological” in Table 1?
What would be the chances of recurrence of VVS in such subjects?
How should such subjects be treated; with just observation or medication?
| LBNP -40 mmHg | 7070° Head-up tilt | |||
|---|---|---|---|---|
| Caucasians | Indians | Indians | Omani Arabs | |
| REF no | 28 | 21 | 11 | 32 |
| HR/min | +18 | +9 | +16 | +19 |
| SBP (mmHg) | -7 | -7 | -1 | -6 |
| DBP (mmHg) | +2 | +3 | +9 | -6 |
| PP (mmHg) | -5 | -12 | -10 | -4 |
LBNP-Lower body negative pressure; HUT-Head-up tilt table; SBP- Systolic blood pressure; DBP-Diastolic blood pressure; PP-Pulse pressure.
Many of these issues are important in those who work in sensitive environments, e.g. pilots. More often than not such findings are enigmatic. During evaluation of Indian Air Force pilots for cosmonaut training (23), one highly experienced test pilot did not have the expected increase in his heart rate and diastolic blood pressure during two 700 HUT tests to which he was subjected on two consecutive days. He was disqualified for the specialized training. Repetition of his HUT tests after three and six months did not reproduce the aberration seen earlier. He had never suffered a VVS during his 18 odd years as a fighter pilot. He was not grounded. As against our observations, Hickler et al (8) found an incidence between 20%-25% in their ot rerwise normal Caucasian subjects exposed to HUT. In the same context, Indian military men did not exhibit any abnormal response to 5 minutes of -50 mmHg LBNP (21) while some of our Caucasian subjects did have mild prodromal signs of syncope when exposed to the same degree of stress (28). Based on these observations, could it be too far fetched to deduce that Indians tolerate orthostatic stress better than Caucasians, possibly because Indians are known to be heat adapted, and hence their physiology for handling body fluid shifts functions more efficiently? Only extensive studies can help to draw definite conclusions on the issue of ethnicity, orthostatic tolerance and syncope incidence. An interesting corollary to this may be that the response of different ethnic groups exposed to orthostatic stress be compared in places like the USA or Singapore where a very wide spectrum of different populations is found in abundance. Inclusion of female subjects will help to resolve the gender differences, if any.
Cardiovascular Response to Orthostatic Stress
When posture is changed from the supine to the erect, about 500 to 600 ml of blood is translocated from the central blood volume (CBV) in the chest to the periphery, mainly the lower limbs because of the normal gravitational effect on the blood column (29). This has been reiterated by many others (1, 15, 28), The orthostatic stress may be applied as postural change from supine to quiet standing (30, 31), HUT, (11, 15-18, 22, 23), or LBNP (6, 19, 21, 28). The well documented response to orthostatic stress consists of a tachycardia, increased diastolic blood pressure, minor changes in systolic blood pressure, increase in peripheral vascular resistance including splanchnic circulation, a decrease in stroke volume and the cardiac output (1, 11, 17, 20, 21, 28). More sophisticated evaluation of cardiovascular response of normal young Omani Arabs to 5 minutes of HUT was done by Jaju (32). While recording online heart rate variability and sympatho-vagal balance (LF/HF) during the HUT in her subjects, she clearly demonstrated that the overall baroreceptor sensitivity (during both up slope and down slope) diminished significantly while the LF/HF ratio increased significantly, the latter clearly indicating heightened sympathetic influence on the cardiovascular system. Increase in sympathetic activity was demonstrated as the highly significant increase in blood norepinephrine (NE). In the same context, incremental muscle sympathetic nerve activity which is a response to sympathetic stimulation has been recorded in human subjects with graded depletion of the CBV [Rea and Wallin (1989)] (33). In summary, a depletion of the CBV requires activation of the sympathetic system which must produce two seminal cardiovascular effects: an increase in heart rate and peripheral vasoconstriction in order to maintain adequate perfusion to the brain in order to avoid LoC. As a consequence, the typical observations include tachycardia, an increase in diastolic blood pressure, minor changes in systolic BP, and a fall in stroke volume, and despite the tachycardia, in the cardiac output because of the lowering of the stroke volume (11, 15, 17).
The Neural Arc for the Baroreceptor Reflex
The receptors which sense the changes in the circulating fluid volume and the arterial blood pressure are the low pressure cardio-pulmonary receptors (34-37), and the high pressure arterial baroreceptors (38-40), respectively. Also to be noted is the presence of endocardial sensory receptors particularly in the left ventricle, sub-served by vagal C fibres which have been associated with producing a bradycardia and hypotension under a set of circumstances which may be relevant to the topic in hand (38-40).
The natural physiological stimulus to the low pressure cardiovascular receptors (type B atrial receptors) is the stretch generated by the circulating fluid volume and hence they are also referred to as volume receptors (35-37). Their activation results mainly in an increase in heart rate (the Bainbridge effect), and a decrease in secretion of the anti diuretic hormone (ADH) by the hypothalamus (35). As against this, a reduction in circulating fluid volume as also a decrease in the CBV which diminishes receptor stretch produces effects which are not diagonally opposite. Under these circumstances, there is an increase in forearm vascular resistance (FVR) without a change in heart rate or blood pressure (34, 37). The effects are accompanied by some increase in plasma catecholamines. These workers (34) subjected volunteers to LBNP of -5 mmHg and observed that the increase in FVR of their subjects had peaked even at this low grade of stress in which only a small depletion in CBV (not measured) would have occurred. Tripathi and Nadel (41) confirmed these findings with their LBNP experiment. In our own series of LBNP exposures, we observed that FVR in our healthy male Caucasian subjects had reached its zenith by the time LBNP of -30 mmHg had been achieved. Thereafter there was no further change in FVR even up to -50 mmHg LBNP stress (32). That sympathetic nerve activity indeed increases with a relatively small depletion of the CBV has been confirmed and has been attributed indisputably to the deactivation of low pressure cardiopulmonary receptors (33). The afferents from these receptors travel up the vagus nerves in both medullated (atrial type B and A), and C fibres to synapse with the second order neurons in the nucleus of the tractus solitarius (NTS). The neurotransmitter released when they are activated is glutamate (40, 42, 43).
Endocardial receptors, particularly those located in the left ventricle have been known to decrease heart rate and vascular tone when stimulated chemically or by mechanical stretch (40). Their C fibre afferents also travel in the vagus nerve to synapse with the 2nd order neurons of the NTS. Excessive contraction of the left ventricle may stimulate these receptors mechanically to produce bradycardia and hypotension (44) seen in subjects with VVS.
By far the most powerful influence is exerted on heart rate and peripheral vascular tone by the arterial baroreceptors located in the carotid sinus and the aortic arch (40). For the purpose of this review which takes in to account the effects of de-activation of baroreceptors, only the effects of the carotid sinus high pressure baroreceptors have been entertained.
The carotid sinus arterial (high pressure) baroreceptors are stretch receptors which respond to pulsatile stretch generated by the systole. They function physiologically between a pressure range of about 50 mmHg to about 200 mmHg. The number of inhibitory impulses sent into the NTS along the IXth cranial nerve peak at the upper limit of pressure, and reach their nadir at 50 mmHg when deactivated (40). The neurotransmitter (glutamate) amount released by the nerve terminals on the NTS neurons is thus reduced if there is a decrease in the pulsatile pressure in the carotid sinus. This must logically happen on reduction in the CBV under various circumstances, most commonly on change of posture from supine to standing to reduce the inhibitory control on the heart and the vasculature with a simultaneous increase in sympathetic activation. The efferent pathway resorted to is either the stimulation or inhibition of the Xth nerve, and reciprocally inhibition or excitation of the sympathetic innervation of the heart and circulation (depending on whether there is activation or deactivation of the receptors).
Central Ramifications of the Reflex and the Neurotransmitters Involved
The baroreceptor afferents synapse with the second order neurons of the NTS. The excitatory neurotransmitter released by these nerve terminals is glutamate (40, 42, 45) which occupies ionotropic glutamate receptors on the NTS (42, 46). The glutamate is released at the afferent nerve terminals and finds its way to the ionotropic receptors on the NTS neurons to stimulate them (Fig. 1). It may be deduced that when the baroreceptors are deactivated (as during reduction in CBV) the volume of glutamate is reduced and vice-a-versa as the release of the glutamate is governed by the frequency of baroreceptor firing (42). As the stimulation of NTS neurons by baroreceptor afferents is responsible for the inhibitory effect on the heart and peripheral circulation, a deactivation of these receptors resulting in a decrease in release of the neurotransmitter will be responsible for releasing the inhibitory effect of the NTS on the heart and peripheral vascular tone. The occupation of ionotropic glutamate receptors on the presynaptic membrane of the NTS neurons is regulated by the generation of metabotropic glutamate receptors (mGluRs) thus making the process self limiting. Of the three types of mGluRs (Type I, II and III), type II are involved in limiting activity of glutamate at the presynaptic 2nd order neurons of the NTS.
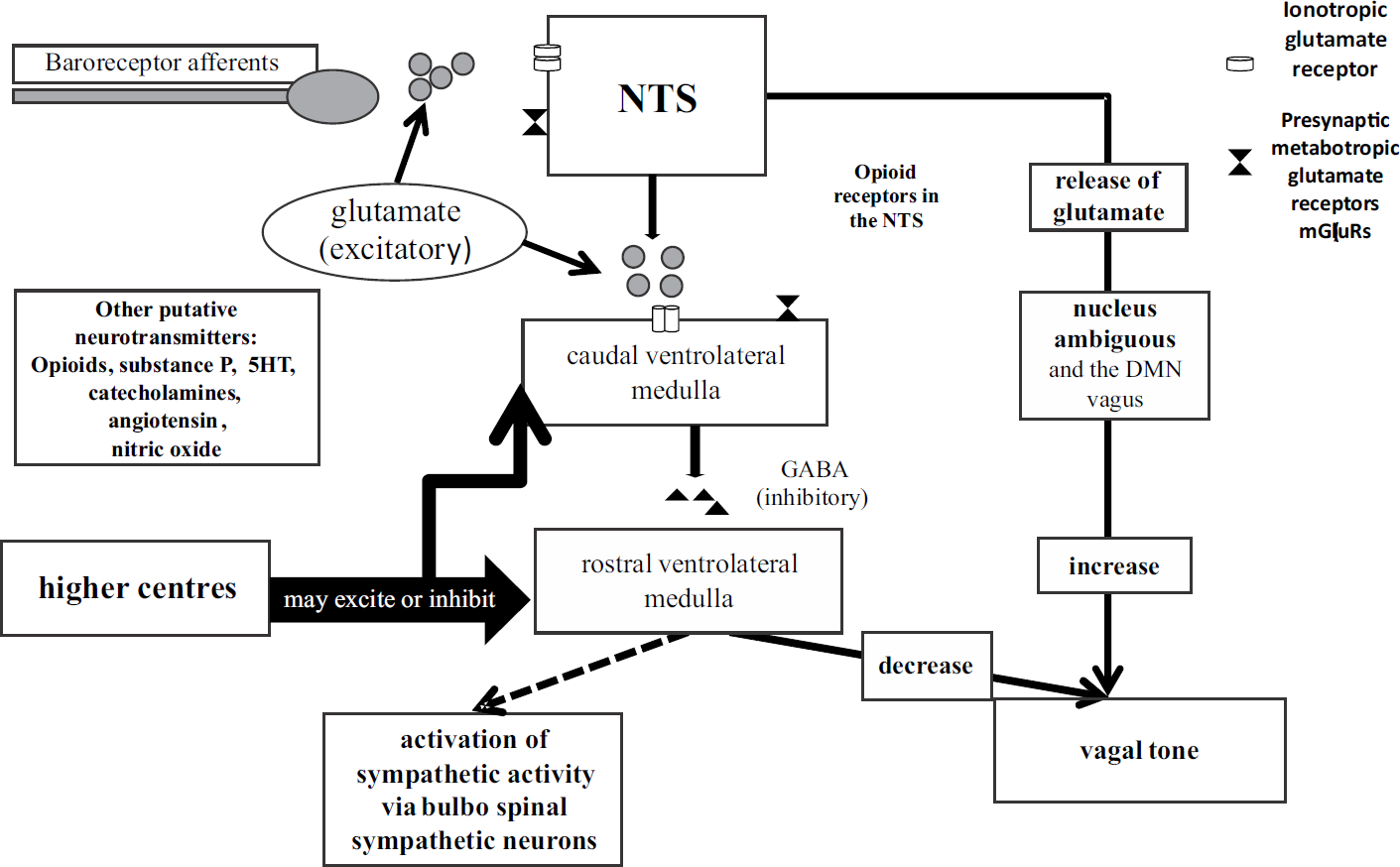
- Central and neurotransmitter interactions in the NTS
- Flow chart constructed on the basis of reference nos. 40, 42, 43, 45 to 48, inhibitory influence is shown by the dotted arrow.
There is evidence that the mGluRs are synthesized in the terminal portion of the afferent nerves synapsing with the NTS (46). The NTS in turn sends excitatory input (with glutamate as neurotransmitter) to the caudal ventrolateral medulla (CVLM) which in turn keeps the rostral ventrolateral medulla (RVLM) inhibited via gama amino butyric acid (GABA) as its transmitter. It has been postulated that this release of GABA in response to stimulation by glutamate from the NTS neurons is self regulated by mGluRs metabolizing the glutamate on the presynaptic membrane of the CVLM (45). The RVLM has a constant excitatory input to the bulbo-spinal sympathetic nerves innervating the heart and peripheral circulation, and inhibits the vagus, but this stimulatory effect is kept under inhibitory control by the CVLM. The NTS also sends excitatory input to the vagal motor nuclei, the Nucleus Ambiguous (NA) being mainly concerned with vagal innervation of the heart (40) and Fig. 1. Therefore, the NTS and its associated inhibitory influence is constantly varying under the influence of glutamate even at rest. If and when the release of glutamate is restricted by a decrease in the afferent input by the various baroreceptors, the inhibitory influence of the NTS reduces because the quantum of excitatory glutamate from the afferent nerve endings reduces. The NTS and the ventrolateral medulla are under influence of various higher centres which include the cortex, the hypothalamus, the limbic system and the raphe nuclei. A number of putative neurotransmitters are thought to be acting in this region. These include catecholamines, angiotensin, opioids, nitric oxide (NO) and substance P (47). Most of the conclusions drawn have been from small animal studies (42, 45-47). Opioid receptors are found in abundance in the NTS and it is the mu-type receptor of the available kappa, delta and mu-opioid receptors that has been linked to excitatory cardiovascular effect (48). That endogenously produced opioids may have a role in humans at the level of the NTS was suggested by Schobel et al (49) when they demonstrated in normal subjects that naloxone, an opioid receptor antagonist, enhanced a low pressure cardiovascular receptor induced reflex. But contrary to this Wallbridge et al (50) refuted the idea of endogenous opioids playing a role in cardiovascular control as they reported that their patients of recurrent syncope did not benefit from naloxone. Thus, endogenous peptides, opioid receptors and their role in central interactions at the level of the NTS are again confounding. The overall understanding though is that opioids influence the NTS in producing a cardio-inhibitory effect and this effect may be suitably neutralized by use of naloxone, an opioid blocker.
The NTS has serotonergic neurons which modulate cardiovascular response to a variety of stresses via a multiplicity of 5HT receptor types (51). Blockage of 5HT1A and 5HT3 receptors reduces the brain stem 5HT uptake increasing the local levels of the neurotransmitter. This may induce a down regulation of post synaptic 5HT receptors (52) in the dorsal motor nucleus of the vagus where 5HT acts to produce bradycardia of baroreceptor stimulation. Knowledge of the role that 5HT plays as a neurotransmitter in the NTS/NA of the vagus is, therefore, confusing at best. The overall central interactions of the NTS and associated medullary neurons with the cortex, hypothalamus and the limbic system must be kept in mind while considering pathophysiology of “VVS or the common faint” which is addressed by this review.
A number of neurotransmitters in peripheral circulation need to be mentioned. Post ganglionic sympathetic NE is released at the sympathetic nerve endings and is mainly responsible for vasoconstriction of the veins and the arterioles (40). Application of even low degree of LBNP was accompanied by significantly raised blood concentration of catecholamines (34). A sudden withdrawal of sympathetic activity will thus be accompanied by a sudden depletion of catecholamines available for contraction of the vascular smooth muscle and produce a dilatation. NO is a local chemo-transmitter produced by endothelial cells of the arteries and arterioles and is a powerful vasodilator (40). It is synthesized from L- arginine by endothelial nitric oxide synthase (eNOS) because of the shear stress exerted on the endothelial cells by the blood flow. During sympathetic activation, the blood flow velocity may increase to generate shear stress which in turn increases the NO production in order to regulate the degree of vasoconstriction that would otherwise be brought about by sympathetic excitation (53). NO produces relaxation of vascular smooth muscle by producing cyclic GMP which in turn generates various protein kinases which expel intra cellular Ca++ from the smooth muscle cell, thus helping the process of dilation (54). Selective excessive synthesis of NO which may produce large scale vasodilatation causing hypotension as in VVS has not yet been reported, but excess synthesis of NO in patients of severe shock where in which large scale vasodilatation occurs is known (55).
A powerful peptide, endothelin, is expressed by endothelial cells of arterioles (40) and secreted as a local hormone taken up by endothelin receptors (ETA ) on the vascular smooth muscle. This normally opposes the vasodilatation being produced by the NO also being secreted by the endothelium (56). Physiologically therefore, there is a continuous to-and-fro battle being waged locally between endothelin which vasoconstricts the vascular smooth muscle and NO which does the opposite. Endothelin by itself may self regulate its vasoconstrictor effect by occupying ETB receptors which, though numerically much less in number than the ETA receptors, are present on the vascular smooth muscle in some vascular beds. The ETB receptors then help in the synthesis of NO by the smooth muscle cells.
Clinical Manifestations of Vasovagal Syncope and Factors Aggravating it
Most reports describing presentation of syncope have encountered symptoms and signs listed in Table 3 (8-11, 57). The episode often start as a prodrome with mild symptoms such as restlessness, visual blurring, sweating and may progress in severity to a LoC. Subjects have been known to slip directly into a LoC without the prodromal symptoms. The percentage reported for this is as high as 39% in patients who had developed the malady (58). Rarely convulsions may occur as a part of VVS (59, 60) though we have not observed any such in our subjects who developed syncope during HUT test or LBNP exposures. Recovery is spontaneous when the subject becomes supine after the LoC, and it is in his/her best interest that alacrity is not shown by care givers in putting the person upright. Some post syncope symptoms such as fatigue, confusion, disorientation have been reported in as much as 76% of patients (58). Such symptoms have been known to last for about 15-20 minutes in the recovery period (61). A family history is often forthcoming in syncope patients (58) and should be enquired into while evaluating an individual with syncope. There are two possibilities when the simple faint may prove to be dangerous. An awkward fall because of the LoC may cause a serious injury especially in the elderly. In fact a fairly high percentage of VVS patients have been reported to have had soft tissue injuries (58). In our experience, the few subjects who developed syncope or presyncopal symptoms did not have a fall or injuries, as they were in a laboratory situation and were promptly attended to. Patients who have had episodes of mild vertigo should also be tested with HUT as the accompanying dizziness could be a symptom of syncope (11). If syncope occurs in an individual in charge of a moving vehicle (e.g., an aeroplane in flight), a serious accident may result because of the ensuing loss of control. A VVS may be precipitated because of environmental factors such as fatigue, insomnia, dehydration, exposure to severe heat stress, painful conditions, medications and anxiety (Table 1). It is imperative, therefore, that a detailed history is obtained in a patient to ascertain if any such factors could have contributed to the episode. This is relevant in two ways. It helps to distinguish between a one off episode and recurrent syncope, and in advising the sufferer as to the precautions that need be taken in order to avoid recurrence. In aircrew a more considered view may be taken while re-flighting if the s/he has been subjected to environmental /situational stress/s as listed.
| 1. | Giddiness/light headedness |
| 2. | Dizziness |
| 3. | Restlessness/nervousness |
| 4. | Headache/nausea/vomiting |
| 5. | Abdominal discomfort |
| 6. | Copious yawning |
| 7. | Sweating |
| 8. | Palpitations |
| 9. | Blurring of vision |
| 10. | Confusion |
| 11. | Pallor |
| 12. | Loss of consciousness without any of the above |
The most prominent cardiovascular finding in a person who has developed LoC as a result of a VVS is a significant bradycardia. The precipitous fall in the blood pressure is often difficult to record as there is more often than not a time gap between the occurrence of the episode and the first BP measurement. By the time regular measurements are established the recovery is underway with regaining of consciousness. It is imperative that a patient of VVS is evaluated using on line monitoring technology (Fig. 2). Without it, it is almost impossible to draw any definite conclusions about the condition in the given patient. The BP and heart rate take a variable time to come back to normal, the BP usually taking longer as compared with the heart rate.

- On line record of a young male subject going in to VVS during HUT.
- (Courtesy HOD Physiology and Clinical Physiology, College of Medicine, SQU, Muscat, Oman)
- The arrows show the steep fall in the given parameters with the VVS. Note that the decline in the TPR and the BP started even as the tachycardia was accentuating indicating that the hypotension was initially because of the vasodilation. Despite this the stroke volume and the cardiac out put were maintained by the tachycardia.
Differential Diagnosis
In the differential diagnosis it is important to distinguish between VVS and the postural tachycardia syndrome (POTS) which again is more commonly seen in females. The symptoms of the latter condition are often quite similar to the prodrome of VVS except that LoC is a rare occurrence during POTS. Tachycardia, exercise intolerance, lightheadedness (dizziness), extreme fatigue, headache, blurring of vision are usually noted. In the upright posture, the heart rate increases by >30 /min within 5 to 20 minutes of the orthostatic stress while the blood pressure is maintained. The blood NE is markedly elevated to > 600 pg/ml. The symptoms tend to disappear quickly when the subject resumes supine position. Exposure to heat stress, dehydration, and anxiety inducing situations promote its onset (62, 63). The differences between the two conditions become particularly relevant in the Armed Forces personnel who are commonly exposed to various environmental stresses which are known to precipitate either. VVS is relatively innocuous and is not known to be associated with a permanent autonomic abnormality as such, while in POTS one of the possibilities is the permanent inadequacy of lower limb adrenergic innervation, be it because of antibodies to the alpha1 receptors in the blood vessels or a genetic deficiency of NE transporters (63). On the other hand in VVS and /or orthostatic intolerance, response to orthostatic stress is only temporarily affected in people with an otherwise normal autonomic nervous system (64). It may be noted here that authors sometimes segregate VVS from orthostatic hypotension (65) while for the purpose of this review no such differentiation is being made while trying to understand this knotted issue.
Pathophysiology
The two cardinal features noted at the time of a VVS are: i) a decrease in peripheral vascular tone; and ii) bradycardia. This combination rapidly induces hypotension, reduces brain perfusion which, if it persists for a period of about 8 to 10 seconds, will result in LoC. This is in obvious contravention to the tachycardia and peripheral vasoconstriction that must normally occur as a reflex on change of posture from supine to erect in order to maintain brain perfusion and normal activity. It may be, therefore, helpful if the issue is analyzed under the two separate heads listed earlier.
As seen in Fig. 2, an initial decline of the peripheral resistance followed by a relatively rapid fall precedes the onset of bradycardia in a presyncopal/syncopal episode. A reflex peripheral vasoconstriction is produced by sympathetic activation because of deactivation of the arterial baroreceptors. This should normally help to maintain the volume availability/volume capacity ratio, and hence the venous return under circumstances which tend to cause peripheral vascular pooling (5). This is supported by experiments in which recording of muscle sympathetic nerve activity (MSNA) was shown to increase in response to peripheral vascular pooling (33, 34). Vadaddi et al (66) too reported an increase in MSNA with increasing depletion of the central blood volume. But what was intriguing was their observation that this happened not only in their normal subjects who did not develop syncope, but also in their patients of recurrent VVS. That MSNA activity was withdrawn at the time of presyncope/ syncope has been reported by (67). Therefore, widespread peripheral pooling including that in the splanchnic vessels which occurs at VVS is secondary to the withdrawal of sympathetic vasoconstrictor support to the peripheral vasculature. Mosqueda-Garcia et al (68) on the other hand reported that in patients of recurrent syncope, the decrease in baro-reflex sensitivity (BRS) and NE availability was accompanied by a near total disappearance of MSNA. It is difficult to explain the contrasts in the findings of Vadaddi et al, and Mosqueda-Garcia (66, 68). One possibility which may do so is the timing of recording of the MSNA during the episodes.
In a normal situation, the neurotransmitter NE in this case is released at the nerve terminals on receiving excitatory nerve signals. Some of it occupies the adrenergic receptors to produce vasoconstriction while some spills over into the plasma (and can be measured). Most of the unused NE is picked up by the NE transporters (NET) and repacked in to the NE vesicles at the nerve endings for subsequent use. Also available in the cytoplasm of the vesicle is a Vesicular Monoamine Transferase which sends the NE collected in the cytoplasm to the vesicles which are being recharged by a protein Dynamin (66). These authors (66) went further to demonstrate that in patients who were diagnosed with recurrent VVS, the availability of NE at the sympathetic nerve terminals was significantly reduced despite exaggerated sympathetic nerve activity as recorded by microneurography. They attributed this to reduced presence of tyrosine hydroxylase at the nerve terminals. This enzyme catalyses the conversion of tyrosine to DOPA which progresses further to form NE via dopamine. As against this, in normal subjects, there appeared to be enough NE spill over and adequate availability of NET molecules. In the same context, it has been suggested that one possibility in the pathophysiology of POTS is a deficiency of NET in the lower limb vasculature (65). Carrying the argument further, it may be hypothesized that reduced availability of the GTPase (Dynamin) at the nerve terminals will result in reduced number of vesicles for NE and hence could be responsible for limited availability of NE when required despite sufficient volleys of sympathetic nerve impulses, creating another situation of dysautonomia. To date, we are unaware of objective evidence for this hypothesis.
Reflecting on the evidence cited so far, it may be concluded that in normal individuals, and perhaps in those who have an occasional /incidental syncope, the response to central blood volume depletion is a strong sympathetic response indicated by an increase in the MSNA with spill over of NE into circulation. At the time of syncope, this sympathetic activity is suddenly withdrawn, leading to wide spread vasodilation which drastically reduces venous return, causing hypotension, and diminished brain perfusion. As against this, the peripheral vasodilatation preceding syncope in patients with recurrent syncope seem to behave differently in that despite high degree of MSNA, the NE availability at the adrenergic receptor level is compromised, and this produces the vasodilatation which contributes to the compromised brain perfusion. The above findings may be taken as sufficient evidence that pathophysiology of recurrent VVS (and POTS) is very clearly a dysautnomia, not an innocuous condition (namely an incidental VVS) in which there is a temporary loss of cardiovascular control followed by complete recovery. A conclusion that the former disability(s) exist (s) in an individual may be arrived at by obtaining a detailed clinical history. Laboratory investigations will clinch the diagnosis. Those diagnosed with these melodies, therefore, must be declared permanently unfit for professions such as flying.
The evidence thus far suggests that at the time of occurrence of the faint, there is a sudden reduction in peripheral vascular tone brought about by a withdrawal of sympathetic vasoconstriction. What then is responsible for this vasoconstriction withdrawal? Bechir et al (69) reported that BRS (ms/mmHg) calculated using Fast Fourier Transformation was significantly lower in subjects who had suffered VVS as compared with normal subjects, at rest as well as during central blood volume depletion by LBNP. They argued that it was responsible for relatively poor MSNA activity during central blood volume depletion in these subjects of VVS, though this may not be the only cause and effect relationship. Arguing against this, Furlan et al (70) commented that “Baroreflex function can be depressed by suprabulbar central influences and also by vagal, somatic, or sympathetic afferents”. (Perhaps a more appropriate expression could be Baro-reflex dysfunction rather than depression). The complex interactions around the NTS have been described earlier. For the withdrawal of the vasoconstrictor tone which should be enhanced during CBV depletion in order to maintain the volume availability/volume capacity ratio close to normal, activity of the CVLM needs to be enhanced (Fig. 1). This in fact is in contravention to the normal physiological role of this nucleus which is supposed to be activated by increased baroreceptor afferent input, not during reduced input which is the case under these circumstances. In that case, an influence other than the CVLM must directly inhibit the RVLM (thus rapidly withdrawing the sympathetic excitatory response) or excite the CVLM which in turn would increase its inhibition of the RVLM. Perhaps higher central influences (Fig. 1) play this decisive role. Recently, advanced methods in neuroimaging have been used to study subjects who had common faints as against those who did not have such a history. Beacher et al (71) found that the medullary and mid brain volumes were reduced in patients who had had syncope while Kim et al (72) using voxel-based morphometry and regional volumetry, reported smaller volumes of insular cortex in VVS patients. An extensive review has further suggested that the frontal lobe has connections with the NTS and the hypothalamus, and hence any dysfunction in the former may affect sympathetic outflow (73). In that review, the authors have opined that cortical thickness of those who have common syncope was different to those who suffered from POTS. All these reports suggest a complex higher centre dysfunction in patients with VVS and allied maladies. Does than this demands that based on radiographic findings, ALL those who have suffered a syncope must be dubbed, anatomically and physiologically abnormal? If so even those who have reported VVS brought about in the presence of situational disturbances (Table 1) must be labeled as permanent suspects. At this point of time, however, such a conclusion seems too far fetched to be implemented, especially in specific professions such as aviation. It may also be impractical to evaluate each and very person with VVS in such a detailed manner to determine his/her fallibility as physiologically normal.
NO is a well established endothelial relaxation factor (EDRF) responsible for arteriolar dilatation (40). Normally, there is a reciprocal control between vasoconstriction (adrenergic) and vasodilation (NO induced) in the peripheral vasculature which helps to maintain adequate tissue perfusion at all times. During CBV depletion, in order to maintain blood pressure, it is the vasoconstrictor element which must dominate in order to maintain diastolic blood pressure (arteriolar constriction), and support venous return by inducing venoconstriction. But if NO induced vasodilation overcomes the adrenergic vasoconstrictor effect, the resulting vasodilation will contribute to a possible VVS. That this happens in young adults with recurrent VVS was elegantly demonstrated by Stewart et al (74). They concluded that in patients with recurrent VVS, there is large scale vasodilation, particularly in the splanchnic bed which is mediated by NO which is secreted by neuronal NO via nitrergic nerves, a nomenclature suggested by Toda et al (75) to describe neuronal NO synthesized in postganglionic parasympathetic nerves to distinguish them from cholinergic nerves. In their subjects Stewart et al were able to reverse the vasodilation by administering NO synthase inhibitors, and in fact have suggested that this may be a valid therapeutic approach in treating patients of recurrent VVS.
It has been mentioned earlier that endothelin produced locally by endothelial cells help to generate NO by influencing ETB receptors while at the same time producing vasoconstriction by occupying ETA receptors (56). It may be hypothesized that in some individuals prone to recurrent VVS, the ETB receptors overwhelm the ETA receptors numerically. This may then make the endothelin released to induce more NO secretion, and hence vasodilatation, in lieu of its expected physiological action of vasoconstriction.
These arguments may explain the pathophysiology of recurrent VVS to an extent. But what about the occasional/incidental syncope in otherwise normal individuals? Could there be other mechanisms which induce such events? During sympathetic stimulation, there is an increase in velocity of blood flow. This induces shear stress on the endothelial cells, prompting them to generate NO (40). It could be hypothesized that in otherwise normal individuals who develop a VVS under a set of environmental / situational stresses (Table 1), the shear effect produced by the stress may be of such a high order that the NO generated may be quantitatively enough to overcome adrenergic vasoconstriction and cause a peripheral pooling which is a preamble to progressive decrease in the venous return. Not only are the peripheral effects of NO relevant in cardiovascular control, but as argued by Zanzinger (76), NO has an important role in manipulation of peripheral vascular tone by interfering with sympathetic activation of peripheral circulation at the level of the NTS and its ramifications to provoke pathophysiological vasodilation which progresses to VVS. The above discussion thus brings out emphatically the role of uncontrolled vasodilation, whether because of withdrawal of sympathetic vasoconstriction, or because of actions of NO at the centre or at the periphery, as a major contributor to the pathophysiology of VVS.
The bradycardia of VVS is the other major contender in the causation of VVS, and in all probabilities is reflex in origin. The most consistent explanation for the reflex bradycardia is that it is probably a response to excessive stimulation of the ventricular mechanoreceptors chemically or mechanically (the Bezold-Jarisch reflex). This occurs because the “near empty ventricles” beat vigorously to stimulate the mechanoreceptors in their walls. The afferents are carried by vagal C fibres to the NTS from where they orchestrate the bradycardia (38, 44, 57) to set up the cardio depressor part of the reflex. (The latter's assertion that mechanoreceptors which affect the heart rate are to be found in the urinary bladder, the intestines, and the lungs may help to explain the syncope which may be brought about by micturition, defecation and a bout of coughing).
Certain factors such as prolonged standing (even in normal individuals), exposure to heat stress, use of vasodilator medications etc tend to promote the venous pooling. The hydrostatic pressure exerted by the peripheral pooling would accentuate the loss of fluid into the interstitial space, further compromising the venous return. Greater this loss, more is the sympathetic nerve activity as measured by microneurographic recording techniques (66, 67). It is conceivable, therefore, that exaggerated sympathetic activity occurs. This in turn will produce powerful contractions of the ventricles which as such are relatively “empty” because of the diminishing venous return, and thus precipitate the bradycardia adding to the already established lowered vascular tone. VVS could be the outcome. This hypothesis supported by Yamanouchi et al (77) who found that in their subjects with a history of VVS, the left ventricular (LV) volume as measured using 2D echocardiography reduced quite significantly during HUT while that of their normal subjects did not. They carried these findings forward by suggesting that it may indeed be the very low LV volume during CBV depletion that may precipitate the reflex bradycardia seen during VVS. Surprisingly, however Novak et al (78) have refuted this theory by demonstrating that the ventricular chamber size measured using echocardiography did not change during a presyncope or a frank syncope. In another study, eight healthy young men were depleted of their CBV using LBNP until they reached presyncopal level. 2D echocardiography at this stage showed only a 28% decrease in LV volume, not a near empty ventricle. On the basis of these findings the authors concluded that Bezold Jarisch reflex is not brought into play when VVS occurs (79). Perhaps if the degree of stress applied by them was severe enough to precipitate a frank syncope, they may have found “near empty” ventricles. There is therefore an ambiguity in the occurrence of a significant bradycardia as a pathophysiologic event during VVS. That the Bezold Jarisch reflex is responsible for the syncopal bradycardia is indeed apocryphal has been suggested by Fitzpatrick et al (80). Some of their heart transplant patients developed near syncopal episodes with bradycardia on exposure to a decrease in CBV. They have argued that this suggested partial vagal innervation of the transplanted donor heart, but there was no evidence to suggest that these hearts could have developed afferents from the LV mechanoreceptors which are necessary to bring on the Bezold Jarisch reflex. Nevertheless, in the classical VVS scenario (Fig. 2), there is no doubt that bradycardia forms an important event in the progression of VVS. This conclusion is strengthened by the findings of a study by Santini et al (81) who were able to prevent the bradycardia in their subjects who developed syncope during HUT exposure with intra venous administration of atropine to block post ganglionic muscarinic cholinergic receptors. In conclusion, bradycardia is an integral part of VVS. As to whether it is induced because of the Bezold Jarisch reflex or because of the complex interactions in the region of the NTS is a matter of debate.
Baro Reflex Sensitivity (BRS) and Syncope
“BRS is defined as the change in inter beat interval (IBI) in milliseconds per unit change in BP” (82). In principle, it indicates as to how the sinus code autonomic innervation reacts to oscillations in the blood pressure on a beat to beat basis. This has also been referred to as cardio-vagal BRS (83). Jaju (32) using noninvasive online recording of blood pressure and ecg (RR intervals) computed the BRS in normal young Omani Arabs using the Sequence method. In this, the computer identifies a minimum of four consecutive beats with either a widening of their R-R intervals (Up events) or with a reduction in their R-R intervals (down events), and calculates the change in the time (msecs) per 1 mmHg change in the systolic BP. She reported that the BRS reduces from about 24 msec/mmHg to about 10 msec/mmHg at the end of 5 minutes of 700 HUT indicating a sympathetic dominance on the SA node. Others have also reported similar figures in normal subjects (83). The BRS has been found to be low even in resting conditions in those with poor orthostatic tolerance and the index does not change much on exposure to orthostatic stress (69, 84). In fact it has been suggested that the defect may lie at the sensory receptor level where the changes in the pulsatile pressure are not sensed adequately (69). Surprisingly though the same authors (69) do not report a decrease in the BRS of their normal subjects during orthostatic stress. A low resting BRS does not augur well for SA node nervous control, and has been associated with serious heart disease and hypertension (82, 85, 86). On the other hand, not much difference was noted in the BRS of normal subjects and patients of VVS at rest or during HUT (Table 4). How does one then use BRS in the assessment of VVS subjects, and how relevant is it? Is it best to leave BRS as a prognostic measure in serious heart disease rather than link its presence to the so called innocuous VVS? Theoretically sufferers from an occasional VVS at the time of evaluation should have a normal robust BRS which decreases during HUT, and recovers rapidly to the prepostural change status later as in normal people who have never had a syncope (Fig. 3). That those with recurrent syncope and poor orthostatic tolerance have a low BRS value which does not change much during HUT has already been established (69, 83). Where does that leave the so called “Normal” individual who may have had an incidental VVS probably precipitated by a series of environmental factors in terms of his/her cardiovascular reflex status? Perhaps if such people are subjected to prolonged orthostatic stress (which should produce a syncope in most) and their parameters recorded (including the cardio-vagal BRS), their response and recovery after the VVS episode would closely resemble the so called normal (Fig. 3), while those with conditions such as recurrent VVS/POTS would show a different pattern. A study by Cheng et al (85) seems to support this view. The BRS of their normal subjects and patients of VVS were about 24 msec/mmHg and 22 msec/mmHg. This parameter reduced in both groups when exposed to HUT. While the post tilt BRS of normal subjects recovered to near pre-tilt value, that of the VVS subjects continued to remain low. Such an investigation is likely to help in reflighting of highly trained aircrew who may have had a VVS and have been grounded on the mere suspicion that such an event may recur while in flight and lead to a catastrophic event. Alternatively use of BRS may be confined for use in patients with heart disease such as myocardial infarction, hypertension where a low BRS has been associated with high morbidity/mortality (82, 86, 87).

- Response of a normal healthy individual to change of posture from supine to standing. Note the robust BRS at rest (last tracing) which reduces during standing, and recovers to near original level when back to supine.
- (Record courtesy HoD Physiol and Clin Physiol, CoM,SQU, Muscat, Oman).
| Normal | +ve tilt | ||||
|---|---|---|---|---|---|
| Gender/age | BRS rest | BRS Tilt change | Gender/age | BRS rest | BRS Tilt change |
| F 40 | 17.2 | 11.2 -6.0 | M31 | 6.8 | 7.0 +0.2 |
| F69 | 20.5 | 11.5 -9.0 | F45 | 12.9 | 10.36 -2.54 |
| M57 | 6.2 | 4.54 -1.7 | M33 | -- | 5.3 |
| F55 | 4.4 | 8.48 +4.08 | M27 | 20.2 | 18.14 -1.8 |
| F39 | 12.6 | 12.42 -0.18 | M33 | 12.3 | 9.2 -3.1 |
| F36 | 6.1 | 4.9 -1.2 | F43 | 7 | 9.3 +2.3 |
| F54 | 9.2 | 7.32 -1.88 | |||
(Data courtesy HoD Physiology and Clin. Physiol. CoM and SQUH, SQU).
Thus far two major aspects which contribute to the pathophysiology of VVS have been dealt with: namely the peripheral vasodilation and the bradycardia. However these cannot be looked at in isolation.
Hormonal secretions change during orthostatic stress and alter further if a VVS occurs. A number of hormones are released when CBV reduces (as during applied orthostatic stress). Jaju (32) showed that plasma NE in the blood of her normal subjects increased significantly after 5 minutes of HUT exposure, and this correlated well with autonomic parameters of sympathetic excitation namely LF nu (low frequency normalized units) and LF/HF ratio. In patients who suffer recurrent syncope, plasma epinephrine levels are much higher at near syncope than normal subjects and contribute to the greater degree of vasodilation by co unteracting the s ymp athetic vasoconstriction, and to the more vigorous ventricular contractions as shown by greater LV shortening fraction (which may stimulate cardiac mechanoreceptors) (88). Epinephrine may also cause the over bearing dilation in muscle vascular beds by stimulating beta adrenergic receptors. Others have found that plasma catecholamines, ACTH, Arginine vasopressin (AVP), plasma renin activity and aldosterone increase in normal subjects in whom near VVS is precipitated by using a combination of HUT and LBNP (Jacob et al 1998) (89). Plasma cortisol and prolactin levels increased in those who developed VVS during HUT, the change being attributed to activation of serotonergic neurons in the region of the NTS, show casing the latter as the main objective of study (90). The role of cortisol as a stress hormone during VVS is understandable, but in the current context the conjoint increase in prolactin has been used only to high light the serotonergic neuron activity in the brain, and not for any cardiovascular effects of prolactin. Jardine et al (91) report simultaneous increases in blood AVP and atrial natriuretic peptide (ANP) in their patients of recurring syncope during HUT. They opined that the vasodilation at the time of VVS could have been contributed to the increase in ANP. This is difficult to reconcile with as the natural stimulus for ANP secretion is an increase in atrial filling, not a reduction in heart volumes which occurs with lowering of the CBV. During VVS, the atrial volume would be further depleted, thereby removing the natural stimulus for ANP release. The arguments thus far have shown that the reduced vascular tone and cardiac depression which occur at syncope are interwoven with endocrine changes which are probably triggered by the central ramifications in the region of the NTS, the CVLM and the RVLM, whether it be inhibition of sympathetic vasoconstriction, or stimulation of the vagus, or a combination of both.
Post Space Flight Orthostatic Intolerance
A significant percentage of astronauts /cosmonauts returning to earth after space flight (micro gravity environment) are known to develop symptoms and signs of orthostatic intolerance amounting to presyncope. Full spontaneous recovery occurs in a few days. Frank VVS is uncommon. Disturbance in baro- reflex activity in terms of gain in the baro-vagal reflex has been suggested as the root cause (92, 93). A detailed investigation was carried in 13 astronauts before, during and post space flight (94). In flight, the astronauts themselves performed carotid baroreceptor stimulation and deactivation experiments using neck suction and pressure. The data reflected an increase in sympathetic activity in flight, and not a gain in the baro-vagal reflex as reported by others (92, 93). However, the vagal part probably starts to regain its edge on return to earth and may take as long as a week to re-establish itself. Perhaps on landing the upswing of the vagal influence produces the presyncopal effects such as dizziness and other symptoms and signs. An important contributing factor may be the poor anti-gravity muscle tone because of the zero gravity environment. This may fail to support the peripheral circulation on returning to earth. Other factors suggested are a loss in circulating plasma volume, and vestibular stimulation which result in vomiting and add to fluid loss (94). It is surprising that no mention has been made by any of the authors on the role, if any that ANP may have in the physiological effects of space flight. There is a significant increase in the CBV during space flight and also an increase in the heart volume content (93) as the gravitational effect on the blood column is absent in space. This should act as a powerful stimulus for the release of ANP from the atria, and probably could account for the diuresis observed in the early phase of zero G flight. The high blood levels of ANP may persist on return to earth to cause vasodilation as also the cardio-vagal reflex gain post flight to produce orthostatic intolerance. The only damning factor may be the short half life of ANP (about 2-5 minutes). It may be interesting to study the role of ANP after simulated microgravity conditions produced on earth by water floatation or prolonged bed rest.
The elderly are known to have a high incidence of syncope, especially in the 70 years and more age group (6, 9, 64). It is important to differentiate between the classical VVS and a variety of clinical reasons which may present as a syncope in the elderly, particularly cardiovascular and neurological disorders, and age-induced autonomic dysfunction (Table 1). BRS has been found to decrease with advancing age particularly in the “Down” events, and thus may be a factor which determines the higher incidence of VVS in the elderly (95). VVS is the most common cause of syncope in the elderly and may occur under a variety of circumstances such as change of posture, defecation, coughing, sneezing and micturition. Most patients present with bradycardia as the primary manifestation. What makes it dangerous is the serious injury it may cause to the patient if s/he sustains a fall. Further, the aged are more prone to develop dehydration. They may be consuming medications (diuretics) which may contribute to the dehydration, and lower vascular tone (96). As such this group is handicapped physiologically by a lower circulating fluid volume, making them more susceptible. Another issue to be noted in the care of the elderly is the possibility of their developing VVS after a meal which may induce secretion of vasodilator substances leading to sudden hypotension. Perhaps age induced aberrations in the autonomic nervous system may produce this dysautonomia (14). It is pertinent to note that more often than not, history of the classical prodrome or of past history of having had fainting spells is not forthcoming because of prevalent cognitive deficiencies (57, 97). This makes diagnosis of the condition difficult. Further, the VVS has to be carefully distinguished from other more serious causes of LoC in the elderly namely cardiovascular and neurological events (98). All these issues make VVS a serious proposition in the aged.
Evaluation of VVS
A scrupulous history of the syncopal event is usually enough to distinguish the VVS under discussion here from syncope attributed to various cardiovascular, neurological and other clinical disorders (57, 65, 98). Eliciting information on exposure to various physiological/environmental stresses (Table 1) is essential to accurately attribute the cause of the VVS as it will help to determine as to whether the episode could be classified as “Innocuous”. The information is helpful in the management of such cases by offering advice on preventive avoidance to the stress. It may or may not be essential to further investigate the episode if the history is able to reveal the possible cause. Though in the elderly it may be prudent to use provocative tests to bring on the episode if possible (57). However, we believe that even if there is a single episode of VVS in a specified population such as aircrew, detailed testing is warranted.
Logically the tests that reduce the CBV would be most effective in challenging the cardiovascular baroreceptor reflex mechanism. These include 1. Quiet standing; 2. Head-up tilt table (HUT) test; 3. Lower body negative pressure (LBNP); 4. Combined HUT and LBNP; and 5. HUT + vasodilator drug or LBNP and vasodilator.
Quiet standing is a simple test which does not require special laboratory set up, and can be easily carried out using basic equipment (30), as also using sophisticated monitoring equipment (Fig. 3). Standing dislocates about 400-500 cc of blood from the CBV to the periphery to deactivate the cardiovascular baroreceptors and set up a reparative reflex response. In the POTS, the HR increases by >30 beats /min without any appreciable change in the blood pressure, but with typical symptoms of dizziness, palpitations, sweating after about the 5th minute. LoC is very rare. If a syncope was to occur, there would be symptoms of dizziness, discomfort, a fall in BP followed by a sudden bradycardia and LoC. Repeated measurements need to be made to keep track of the developing situation.
By far the most tried and tested method of evaluating a patient of syncope is the HUT. The degree of tilt can vary from 200 to 700. The latter is most frequently used as hydrostatically it produces the effects of a near vertical tilt (tangent of 700 is close to 1, i.e. of 900) while avoiding the severe discomfort and perception of falling over felt by the subject when tilted by 800 or more. We have used 700 HUT extensively (15-18, 22, 23, 32) to study cardiovascular reflex status as have others (8, 12, 59). The usual duration of HUT stress is 15 to 20 minutes though duration of 5 minutes as well more prolonged (>30 min) have also been used. The longer duration tilts may be used to provoke a syncopal like situation or frank syncope because longer the duration of the stress, greater is the loss of circulating fluid volume into the periphery. The rate of application of the HUT may be maintained constant by using electrically operated HUT table. Ideally it should take about 15 seconds. Faster rate of application may bring about vestibular stimulation which may influence cardiovascular response.
A normal response to HUT consists of an increase in mean arterial pressure, in the diastolic BP and in the in heart rate, and with a fall in the pulse pressure (11). The stroke volume index and the cardiac output index reduce significantly by 33% and 18%, respectively (17). (The stroke volume was measured using LE ejection time method). If HUT induced reflex syncope occurs, the BP starts decreasing after a variable time on assuming HUT. The heart rate (HR) may increase just before syncope and plunges at the time of syncope along with the BP (Fig. 2).
It is difficult to arrive on a consensus regarding sensitivity, specificity and diagnostic yield of the HUT as it depends upon the technique used (99). HUT has acceptable sensitivity and specificity when calculated in patients with true VVS (100, 101). Some authors have recommended a combination of HUT with vasodilator drugs (sublingual nitrates or intra venous infusion of isoproterenol) to provoke VVS during HUT especially in those who may have had an incidental episode. A recent systematic review gives the 66% of overall positivity rate for syncope with nitroglycerine challenge and 61% for isoproterenol challenge (102) . Others opine that drug provocation during tilt helps to reduce the total duration of the tests (103) . Importantly there are those who recommend drug free (non provocative) tilt over pharmacological challenge as it provides better diagnostic yield (100, 101). We too would like to support this view.
When should a HUT be done? We opine that it will be definitely helpful in confirming diagnosis in patients with recurrent syncope and in those apparently normal individuals in selective occupations (pilots) who have had an incidental episode of syncope. Our belief is strengthened by a similar opinion by others (102). It is also useful in evaluating elderly patients with a possible occurrence of VVS because often such patients are unable to give accurate history of the episode (102).
Apart from HUT, we have used lower body negative/suction pressure (LBNP/LBSP) (15, 19-21, 104) to test cardiovascular responses to graded displacement of the CBV in to the periphery. Blood volume displaced by LBNP of -40 mmHg and 700 HUT is similar at about 500-600 ml. The suction may be applied in a graded manner so that the volume of blood displaced increases in a graded manner. This has helped in deciphering the role played by low pressure cardiopulmonary receptors in FVR. At suction pressure below 20 mmHg there is an increase in the FVR without a change in HR, while by -30 mmHg, tachycardia induced by deactivation of arterial baroreceptors sets in without a further change in the FVR (21, 34). There are slight differences in the reflex response to the two types of stresses (Table 2). The greater increases in the tachycardia and the diastolic pressure during HUT (11) are attributed to the natural gravitational effect under which the reflex mechanisms normally operate in day to day life (105) making it a better natural stimulus, while during LBNP (21) the subject is in a horizontal position thus eliminating the change in hydrostatic pressure normally brought about by gravity while in the upright posture. This makes HUT a better tool for investigation of the reflex mechanism under discussion.
A combination of HUT and LBNP (-20 and -40 mmHg for 10 minutes each) has been applied simultaneously to elicit cardiovascular responses to CBV displacement in to the periphery (106). 84% of the subjects who underwent this procedure developed presyncopal effects. The authors argue that this technique is better as the duration of exposure to the stress is markedly reduced. Against this, we would argue that the type of VVS in apparently normal but high risk profession individuals that we are looking at, should fall in to the 16% of those who do not develop any symptoms. This argument seems too farfetched to classify only those in the latter category to be “safe”. We would agree more with those who recommend drug free tilt over pharmacological challenge as it provides better diagnostic yield (100, 101).
Principles of Management of VVS
A detailed approach to management of various types of syncope may be found in the ESC Guidelines for the diagnosis and management of syncope 2018 (100). It may not be necessary to treat vigorously all those with history of VVS (64, 65). A useful approach for those who may suffer from an occasional VVS brought about by environmental situations such as dehydration, prolonged standing, and heat stress exposure would be to ensure that the discrepancy between the availability of circulating fluid volume and the vascular volume capacity ratio is brought back towards as far as possible. This may be easily done by increasing fluid and salt intake as the basic measure. They should be advised on avoidance of such situations in order to prevent recurrence. This should suffice for subjects who are aircrew with a single or occasional history of VVS. The approach should be useful for patients diagnosed with POTS, a distant cousin of the prodrome of VVS. In addition, reassurance and upper torso exercise training has been found to be helpful. As a precautionary measure, such patients are advised to stand up slowly using support. More stringent measures may not be necessary, and if required may be decided upon by the clinician.
In more persistent recurrent VVS, various medications have been tried with variable success. The principle applied was to modulate available knowledge of various neurotransmitters associated with functioning of the NTS. Schobel et al (49) suggested that blocking opiod receptors in the NTS region could be helpful in augmenting the cardiopulmonary deactivation reflex. However, their suggestion was refuted (50). That serotonin may have a role to play in the etiology of VVS was proposed by Jordan (51) and accordingly paroxitine, a selective serotonin uptake inhibitor was tried in the treatment of recurrent syncope with some success (52). However, another analysis by Raj and Koffin did not particularly find paroxitine particularly useful (65). The use of beta blockers was attempted in order to reduce the powerful ventricular contractions which may be responsible for stimulating ventricular mechanoreceptors (69) that form the afferent limb of the Bezold Jarisch reflex. But the results of the use of this class of drugs was not very encouraging (107). Midodrine an alpha adrenergic receptor agonist, has been found to be useful (107). This would help to limit the vasodilation as a major contributor to the pathophysiology of VVS.
The role of NO in causing vasodilation during VVS has been elaborated earlier (74-76). In fact, Stewart et al (74) demonstrated that use of NO synthase inhibitors was helpful in attenuating the vasodilation during of VVS and is deemed promising as a tool for use in patients of recurrent VVS.
There have been attempts to improve responses of patients with recurrent VVS by orthostatic training (84). Patients of VVS were asked to lean against a wall with their feet placed 15 cm away from the wall. Duration of each session was 30 minutes. The authors reported an improvement in 74% of those who completed the training. The success was apparently limited by a relatively low compliance by the volunteers.
Verheyden et al (108) gave daily HUT exposure training to 17 VVS patients of recurrent VVS with the hypothesis that repeated HUT stress will improve vasoconstrictor response. They found that after about 4 exposures the their subjects were free of VVS and prodrome, and they remained symptom free for long as 6 weeks if just quiet standing was continued at home. This could indeed be a useful approach in tackling the problem.
Yoga training affects sympatho-vagal balance positively (109). If VVS is an indication of temporary autonomic dysfunction, practice of yoga should help patients of VVS. That it actually does so was reported by Gunda et al (2015) (110) in female subjects with syncope. Heart rate variability drifts towards vagal dominance signaling alteration in the autonomic control of the sinus node. Theoretically this may increase resting cardio-vagal baro-reflex sensitivity there by helping to alleviate symptoms of presyncope and syncope in these subjects. Our own experience showed that the three cosmonauts (one Indian and two Russian) who flew the joint Indo Soviet space mission in 1983 did not react adversely to orthostatic stress on return to earth (111). They had been trained to do yoga exercises during their preflight training and during the space flight. As to whether it was partly the yoga training that helped them cope with rigors of space flight cannot be ascertained as this was only one such experiment. It may be interesting to give a serious thought to this aspect when ISRO sends up an Indian manned space flight in the near future.
Conclusions
Despite the extensive research and material available on VVS, the entity remains baffling. As to what is a “Safe VVS” especially in certain types of populations such as aircrew still remains partially unanswered. It can be concluded that VVS is a result of a temporary failure of reflex control of the cardiovascular system. The search for the means of overcoming this shortfall on a permanent basis in subjects who suffer VVS is still ongoing.
Conflict of Interests
Nil.
Acknowledgments
Head, Department of Physiology and Clinical Physiology, College of Medicine and Health Sciences, Sultan Qaboos University and Hospital, Muscat, Sultanate of Oman graciously permitted us the use of data given in Table 1 and Figures 2 and 3. Mr. Mohit Dikshit was kind enough to edit the manuscript.
References
- Syncope. An enigma in aviation physiology. In: M Fahim, ed. In: Peripheral Elements of the Nervous System. Delhi: Vallabhbhai Patel Chest Institute; 1996. p. :59-72. Computronix
- [Google Scholar]
- Syncope. Physiological considerations and an analysis of the clinical characteristics in 510 patients. Am J Med. 1961;30:418-438.
- [CrossRef] [PubMed] [Google Scholar]
- Syncope of unknown origin. The need for a more cost- effective approach to its diagnosis evaluation. J Am Med Assoc. 1982;247:2687-2691.
- [CrossRef] [Google Scholar]
- Review article: primary care. Syncope. N Engl J Med. 2000;343:1856-1862.
- [CrossRef] [PubMed] [Google Scholar]
- Sympathetic dysfunction in vasovagal syncope and the postural orthostatic tachycardia syndrome. Front Physiol. 2014;5:290.
- [CrossRef] [Google Scholar]
- Pathophysiology of neurally mediated syncope. Neurolgia. 2016;31:620-627.
- [CrossRef] [Google Scholar]
- Tilt table and plasma volume changes with short term deconditioning experiments. Aerosp Med. 1967;38:564-568.
- [Google Scholar]
- The clinical evaluation of faulty orthostatic mechanisms. Med Clin North Am. 1960;44:1237-1250.
- [CrossRef] [PubMed] [Google Scholar]
- Syncope: epidemiology, etiology, and prognosis. Front Physiol. 2014;5:471.
- [CrossRef] [PubMed] [Google Scholar]
- Incidence of syncope amongst healthy young people. Indian J Physiol Pharmacol. 1988;34(suppl 1):319-320.
- [Google Scholar]
- Orthostatic tolerance of normal Indians and of those with suspected abnormal cardiovascular reflex status. Aviat Space Environ Med. 1986;57:168-173.
- [Google Scholar]
- Head-up tilt testing in children. Eur Heart J. 2001;22:1968-1971.
- [CrossRef] [PubMed] [Google Scholar]
- Vasovagal syncope as a manifestation of an evolutionary selected trait. J Atr Fibrillation. 2014;7:97-102.
- [CrossRef] [Google Scholar]
- Postural stress tests for the clinicophysiological evaluation of cardiovascular reflexes. Indian J Physiol Pharmacol. 1987;31:1-12.
- [Google Scholar]
- Changes in orthostatic tolerance during short duration mild hypoxia. Aviat Med. 1976;20:9-14.
- [Google Scholar]
- Left ventricular ejection time based cardiac output changes in pilots and non-pilots during orthostatic stress. Indian J Med Res. 1986;83:301-303.
- [Google Scholar]
- Systolic time intervals in pilots and non-pilots during 700 head-up tilt. Aviat Space Environ Med. 1986;57:332-335.
- [Google Scholar]
- Immediate and delayed effects of temazepam on human orthostatic reflexes. Br J Clin Pharmacol. 1987;24:254-255.
- [Google Scholar]
- Lower body suction and cardiovascular reflexes: physiological and applied considerations. Indian J Physiol Pharmacol. 1990;34:3-12.
- [Google Scholar]
- Calcium channel blockers and the CVS reflex responses during LBSP. Indian J Physiol Pharmacol. 1991;35:232-236.
- [Google Scholar]
- Comparative cardiovascular response to 70 degrees head up tilt in pilots and nonpilots. Aviat Med. 1983;27:36-42.
- [Google Scholar]
- Medical evaluation of cosmonauts: physiological stress testing. Aviat Med. 1984;28:107-115.
- [Google Scholar]
- Effect of postural changes on cardiovascular parameters across gender. Medicine (Baltimore). 2016;95:e41-e49.
- [CrossRef] [PubMed] [Google Scholar]
- Gender Differences in Cardiovascular Responses to Orthostatic Challenge in Healthy Older Persons. MD Thesis. Med University Graz; 2017.
- [Google Scholar]
- Gender difference in patients with recurrent neurally mediated syncope. Yonsei Med J. 2010;51:499-503.
- [CrossRef] [PubMed] [Google Scholar]
- The cardiovascular response to postural change as a function of race. Biol Psycho. 1995;39:173-186.
- [CrossRef] [PubMed] [Google Scholar]
- Human orthostatic reflexes after taking temazepam at night. Br J Clin Pharmacol. 1987;24:799-807.
- [CrossRef] [PubMed] [Google Scholar]
- The regulation of blood distribution in man. Acta Physiol Scand. 1952;26:312-327.
- [CrossRef] [PubMed] [Google Scholar]
- Autonomic mechanisms in the initial heart rate response to standing. J Appl Physiol Respir Environ Exerc Physiol. 1980;49:809-814.
- [CrossRef] [PubMed] [Google Scholar]
- Heart rate and blood pressure responses of left handers and right handers to autonomic stressors. Indian J Physiol Pharmacol. 2004;48:31-40.
- [Google Scholar]
- Blood Pressure and Cardiovascular Control in Offspring of Hypertensive and Normotensive Parents from Large Homogeneous Omani Arab Pedigrees. PhD Thesis. Graz Teaching Hospital and Institute of Physiology Medical University Graz 2008
- [Google Scholar]
- Sympathetic nerve activity during lower body negative pressure in humans. J Appl Physiol. 1989;66:2778-2781.
- [CrossRef] [PubMed] [Google Scholar]
- Changes in plasma catecholamines in response to reflex modulation of sympathetic vasoconstrictor ton by cardio-pulmonary receptors. Clin Sci. 1985;68:503-510.
- [CrossRef] [PubMed] [Google Scholar]
- Natural stimulation of type B receptors. J Physiol (London). 1963;169:116-136.
- [CrossRef] [PubMed] [Google Scholar]
- Circulatory basis of fluid volume control. Physiol Rev. 1963;48:423-481.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular control from cardiac and pulmonary vascular receptors. Exp Physiol. 2014;99:312-319.
- [CrossRef] [PubMed] [Google Scholar]
- Neurally mediated syncope: a review of cardiac and arterial receptors. J Clin Neurophysiol. 1997;14:170-182.
- [CrossRef] [PubMed] [Google Scholar]
- Reflex control of peripheral circulation. Prog Cardiovasc Dis. 1976;18:371-403.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular regulatory mechanisms, Chapter 32. In: Barrett KE, Barman SM, Boitano S, Brooks HL, eds. In: Ganong's Review of Medical Physiology. New York: McGraw Hill Medical; 2012. p. :587-600.
- [Google Scholar]
- Forearm and muscle vasoconstriction during lower- body negative pressure. J Appl Physiol. 1986;60:1535-1541.
- [CrossRef] [PubMed] [Google Scholar]
- Synaptic transmission in nucleus tractus solitarius is depressed by Group II and III but not Group I presynaptic metabotropic glutamate receptors in rats. J Physiol. 2002;538:773-786.
- [CrossRef] [PubMed] [Google Scholar]
- Neurotransmitters in the nucleus tractus solitarius mediating cardiovascular function. In: Dun NJ, Machado BH, Pilowsky PM, eds. In: Neural Mechanisms of Cardiovascular Regulation. Boston, MA: Springer; 2004. p. :81-98.
- [CrossRef] [Google Scholar]
- Role of cardiac vagal C- fibers in cardiovascular control. Rev Physiol Biochem Pharmacol. 1979;86:1-94.
- [CrossRef] [PubMed] [Google Scholar]
- Glutamate suppresses GABA release via presynaptic metabotropic glutamate receptors at baroreceptor neurones in rats. J Physiol. 2005;562(Pt 2):535-555.
- [CrossRef] [PubMed] [Google Scholar]
- A novel postsynaptic Group II metabotropic glutamate receptor role in modulating baroreceptor signal transmission. J Neuroscience. 2009;29:11807-11816.
- [CrossRef] [PubMed] [Google Scholar]
- Neural regulation of the cardiovascular system, Chapter 36. In: Squire LR, Berg D, Bloom F, du Lac S, Ghosh A, Spitzer N, eds. In: Fundamentals of Neuroscience (3rd). Tokyo: Elsevier; 2008. p. :829-853.
- [Google Scholar]
- mu-Opioid receptors in NTS elicit pressor responses via sympathetic pathways. Am J Physiol. 1987;252:H156-H162.
- [CrossRef] [PubMed] [Google Scholar]
- Naloxone potentiates cardiopulmonary baroreflex sympathetic control in normal humans. Cir Res. 1992;70:172-183.
- [CrossRef] [PubMed] [Google Scholar]
- Role of endogenous opioids and catecholamines in vasovagal syncope. Eur Heart J. 1996;17:1729-1736.
- [CrossRef] [PubMed] [Google Scholar]
- Vagal control of the heart: central serotonergic (5-HT) mechanisms. Exp Physiol. 2004;90:175-181.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of paroxetine hydrochloride, a selective serotonin reuptake inhibitor, on refractory vasovagal syncope: a randomized, double-blind, placebo- controlled study. J Am Coll Cardiol. 1999;33:1227-1230.
- [CrossRef] [PubMed] [Google Scholar]
- The L Arginine-nitric oxide pathway. N Engl J Med. 1993;329:2002-2012.
- [CrossRef] [PubMed] [Google Scholar]
- Nitrite and nitric oxide metabolism in peripheral artery disease. Nitric Oxide. 2012;15:217-222.
- [CrossRef] [PubMed] [Google Scholar]
- Physiopathology of shock. J Emerg Trauma Shock. 2011;4:222-232.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical characteristics of patients with vasovagal reactions presenting as unexplained syncope. Europace. 2001;3:141-146.
- [CrossRef] [PubMed] [Google Scholar]
- Differentiation of convulsive syncope and epilepsy with head-up tilt. Ann Intern Med. 1991;115:871-876.
- [CrossRef] [PubMed] [Google Scholar]
- Convulsive syncope in blood donors. Ann Neurol. 1982;11:525-528.
- [CrossRef] [PubMed] [Google Scholar]
- The postural tachycardia syndrome (POTS): pathophysiology, diagnosis and management. Indian Pacing Electrophyiol J. 2006;6:84-99.
- [Google Scholar]
- Postural tachycardia syndrome: beyond orthostatic intolerance. Curr Neurol Neurosci Rep. 2015;15:60.
- [CrossRef] [PubMed] [Google Scholar]
- The management of vasovagal syncope. Qtly J Med. 2016;109:767-777.
- [CrossRef] [PubMed] [Google Scholar]
- Medical therapy and physical maneuvers in the treatment of the vasovagal syncope and orthostatic hypotension. Prog Cardiovasc Dis. 2013;55:425-433.
- [CrossRef] [PubMed] [Google Scholar]
- Recurrent postural vasovagal syncope: sympathetic nervous system phenotypes. Circ Arrhythm Electrophysiol. 2011;4:711-718.
- [CrossRef] [PubMed] [Google Scholar]
- Sympathetic outflow to muscles during vasovagal syncope. J Auton Nerv Syst. 1982;6:287-291.
- [CrossRef] [PubMed] [Google Scholar]
- Sympathetic and baroreceptor reflex function in neurally mediated syncope evoked by tilt. J Clin Invest. 1997;99:2736-2744.
- [CrossRef] [PubMed] [Google Scholar]
- Dysfunctional baroreflex regulation of sympathetic nerve activity in patients with vasovagal syncope. Circulation. 2003;107:1620-1625.
- [CrossRef] [PubMed] [Google Scholar]
- Baroreflex regulation of sympathetic nerve activity in patients with vasovagal syncope. Circulation. 2004;109:e171.
- [CrossRef] [PubMed] [Google Scholar]
- Vulnerability to simple faints is predicted by regional differences in brain anatomy. Neuroimage. 2009;47:937-945.
- [CrossRef] [PubMed] [Google Scholar]
- Right insular atrophy in neurocardiogenic syncope: a volumetric MRI study. Am J Neuroradiol. 2014;35:113-118.
- [CrossRef] [PubMed] [Google Scholar]
- Alterations of brain network hubs in reflex syncope: evidence from a graph theoretical analysis based on DTI. Brain Behav. 2018;8:e01006.
- [CrossRef] [PubMed] [Google Scholar]
- Nitric oxide synthase inhibition restores orthostatic tolerance in young vasovagal syncope patients. Heart. 2017;103:1711-1718.
- [CrossRef] [PubMed] [Google Scholar]
- The pharmacology of nitric oxide in the peripheral nervous system of blood vessels. Pharmacol Rev. 2003;55:271-324.
- [CrossRef] [PubMed] [Google Scholar]
- Role of nitric oxide in the neural control of cardiovascular function. Cardiovas Res. 1999;43:639-664.
- [CrossRef] [PubMed] [Google Scholar]
- Changes in left ventricular volume during head-up tilt in patients with vasovagal syncope: an echocardiographic study. Am Heart. 1996;131:73-80.
- [CrossRef] [PubMed] [Google Scholar]
- Is the heart “empty” at syncope? J Auton Nerv Syst. 1996;60:83-92.
- [CrossRef] [PubMed] [Google Scholar]
- The heart is not necessarily empty at syncope. Avia Space Environ Med. 1999;70:213-219.
- [Google Scholar]
- Vasovagal reactions may occur after orthotopic heart transplantation. J Am Coll Cardiol. 1993;21:1132-1137.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of atropine in vasovagal syncope induced by head-up tilt testing. Eur Heart J. 1999;20:1745-1751.
- [CrossRef] [PubMed] [Google Scholar]
- Baroreflex sensitivity: mechanisms and measurement. Neth Heart J. 2013;21:58-60.
- [CrossRef] [PubMed] [Google Scholar]
- Differential effect of head-up tilt on cardiovagal and sympathetic baroreflex sensitivity in humans. Exp Physiol. 2003;88:769-774.
- [CrossRef] [PubMed] [Google Scholar]
- Improvement in low upright baroreflex sensitivity is associated with positive clinical effect of orthostatic training. Pacing Clin Electrophysiol. 2018;41:42-49.
- [CrossRef] [PubMed] [Google Scholar]
- Modifications of autonomic activity and baroreceptor response during tilt-induced vasovagal syncope. Computing in Cardiol. 2010;37:943-946.
- [Google Scholar]
- Baroreflex sensitivity and its response to deep breathing predict increase in blood pressure in type 1 diabetes in a 5-year follow-up. Diabetes Care. 2011;34:2424-2430.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical value of baroreflex sensitivity. Neth Heart J. 2013;21:61-63.
- [CrossRef] [PubMed] [Google Scholar]
- False positive head-up tilt: hemodynamic and neurohumoral profile. J Amer Coll Cardiol. 2000;35:188-193.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of standing on neurohumoral responses and plasma volume in healthy subjects. J Appl Physiol. 1998;84:914-921.
- [CrossRef] [PubMed] [Google Scholar]
- Hormonal responses during neurally mediated syncope as an indication of central serotonergic activity. Hellenic J Cardiol. 2004;45:8-13.
- [Google Scholar]
- Neurohormonal response to head-up tilt and its role in vasovagal syncope. Am J Cardiol. 1997;79:1302-1306.
- [CrossRef] [PubMed] [Google Scholar]
- Baroreflex dysfunction induced by microgravity: potential relevance to postflight orthostatic intolerance. Clin Auton Res. 2000;10:269-277.
- [CrossRef] [PubMed] [Google Scholar]
- Dynamic adaptation of cardiac baroreflex sensitivity to prolonged exposure to microgravity: data from a 16- day spaceflight. J Appl Physiol. 2008;105:1569-1575.
- [CrossRef] [PubMed] [Google Scholar]
- Human vagal baroreflex mechanisms in space. J Physiol. 2010;588(Pt 7):1129-1138.
- [CrossRef] [PubMed] [Google Scholar]
- Arterial baroreflex function in older adults with neurocardiogenic syncope. Clin Invest Med. 2013;32:E191-E198.
- [CrossRef] [PubMed] [Google Scholar]
- The management of vasovagal syncope. Qtly J Med. 2016;109:767-773.
- [CrossRef] [PubMed] [Google Scholar]
- Variations in diagnostic yield of head-up tilt test and electrophysiology in groups of patients with syncope of unknown origin. Eur Heart J. 2001;22:857-865.
- [CrossRef] [PubMed] [Google Scholar]
- ESC Guidelines for the diagnosis and management of syncope. Eur Heart J. 2018;39:1883-1948.
- [CrossRef] [PubMed] [Google Scholar]
- Indications, methodology, and classification of results of tilt-table testing. Am J Cardiol. 1999;84:10Q-19Q.
- [CrossRef] [PubMed] [Google Scholar]
- Head-up tilt testing for diagnosing vasovagal syncope: a meta-analysis. Intl J Cardiol. 2013;168:27-35.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of short head-up tilt test with conventional protocol after omission of nonmedicated phase in children and young adults. Adv Biomed Res. 2016;5:207-213.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular reflex adjustments for aerospace application, Chapter 38. In: Manchanda S, Selvamurthy W, Mohan Kumar V, eds. In: Advances in Physiological Sciences. Delhi: Macmillan; 1992. p. :263-271.
- [Google Scholar]
- Comparison of cardiovascular responses between lower body negative pressure and head-up tilt. J Appl Physiol. 2005;98:2081-2086.
- [CrossRef] [PubMed] [Google Scholar]
- Combined head-up tilt and lower body suction: a test of orthostatic tolerance. Clin Auton Res. 1994;41:41-47.
- [CrossRef] [PubMed] [Google Scholar]
- Postural syncope: mechanisms and management. Med J Aust. 2007;187:299-304.
- [CrossRef] [PubMed] [Google Scholar]
- Tilt training increases the vasoconstrictor reserve in patients with neurally mediated syncope evoked by head-up tilt testing. Eur Heart J. 2008;29:1523-1530.
- [CrossRef] [PubMed] [Google Scholar]
- Yoga and heart rate variability: a comprehensive review of literature. Intl J Yoga. 2016;9:97-113.
- [CrossRef] [PubMed] [Google Scholar]
- Role of yoga as an adjunctive therapy in patients with neurocardiogenic syncope: a pilot study. J Interv Card Electrophysiol. 2015;43:105-110.
- [CrossRef] [PubMed] [Google Scholar]





