Translate this page into:
Emerging Diabetic Novel Biomarkers of the 21st Century
Address for correspondence Shilpa Suneja, MD, Department of Biochemistry, Vardhman Mahavir Medical College & Safdarjung Hospital, New Delhi 110029, India (e-mail: shilpasuneja@rediffmail.com).
This article was originally published by Thieme Medical and Scientific Publishers Pvt. Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Diabetes is a growing epidemic with estimated prevalence of infected to reach ~592 million by the year 2035. An effective way to approach is to detect the disease at a very early stage to reduce the complications and improve lifestyle management. Although several traditional biomarkers including glucated hemoglobin, glucated albumin, fructosamine, and 1,5-anhydroglucitol have helped in ease of diagnosis, there is lack of sensitivity and specificity and are inaccurate in certain clinical settings. Thus, search for new and effective biomarkers is a continuous process with an aim of accurate and timely diagnosis. Several novel biomarkers have surged in the present century that are helpful in timely detection of the disease condition. Although it is accepted that a single biomarker will have its inherent limitations, combining several markers will help to identify individuals at high risk of developing prediabetes and eventually its progression to frank diabetes. This review describes the novel biomarkers of the 21st century, both in type 1 and type 2 diabetes mellitus, and their present potential for assessing risk stratification due to insulin resistance that will pave the way for improved clinical outcome.
Keywords
21st century
biomarkers
review
type 1 diabetes mellitus
type 2 diabetes mellitus
Introduction
Diabetes mellitus (DM) is a complex metabolic and multifunctional syndrome that is characterized by persistent hyperglycemia. The prevalence of DM has increased over the past two decades and in coming years, it will be a major health problem for the world.1 According to the International Diabetes Federation report, prevalence of diabetes is spiraling globally with an estimated 425 million adults having diabetes in 2017 and will be increased to ~592 million by the year 2035.2 Developing countries like India will have ~109 million of affected people, making diabetes no more a developed world disease.
DM has a multifaceted pathogenesis that occurs either due to impaired insulin secretion or due to development of insulin resistance (IR) at target tissues or because of insulin deficiency due to autoimmune destruction of pancreatic β-cells.3
DM is classified into different types based on the pathogenic mechanisms as type 1, type 2, gestational diabetes, and other types in which specific, genetic defects, metabolic, and mitochondrial abnormalities and some conditions that impair glucose tolerance are included.4
Diabetes, if untreated, may lead to several serious chronic complications involving microvascular complications like nephropathy or retinopathy and macrovascular complications like cardiovascular disease (CVD) and stroke. The most common cause of mortality in diabetes is, however, macrovascular complications.5
Given the burden of diabetes and its complications, much attention has been given to prevention, beginning with identifying at-risk individuals prior to diagnosis that led to the designation of “prediabetes,” an intermediate form of dysglycemia on a spectrum ranging from normal to overt diabetes.6 According to Centers for Disease Control, one out of three adults had prediabetes and agonizingly, 90% were unaware of their diagnosis.7
Awareness and knowledge regarding the diabetic condition, its management, complications, and risk factors are crucial steps for better quality of life and its control.8
However, the clinical manifestations especially in type 2 diabetes mellitus (T2DM) are delayed by years, thereby restricting its timely diagnosis. It has been demonstrated that there is a significant relative risk reduction of CVDs and all-cause mortality in individuals undergoing regular medical examination emphasizing the importance of early diagnosis of the disease.9
The efficacy of diabetic management to delay progression of DM would be improved if they could be implemented during the initial phases of the disease and number of studies also suggest that progression of diabetes can be postponed or prevented with earlier initiation of current treatment protocols.10,11 Thus, the prediction and early identification of high-risk individuals before the onset of prediabetes stage, that is, when the β-cells are relatively intact, along with timely identification of diabetic complications, are of paramount importance, for effective intervention. Timely management though is helpful in preventing progression to overt disease but, however, remains a challenging scenario.
Considering the need of present situation, prompt diagnosis and innovation of an effective and safe treatment option are required to achieve this goal, identifying new biomarkers for predicting individuals at high risk of diabetes and its complications have, therefore, become a priority for targeting preventive measures efficiently.12,13
Thus, this review is intended to discuss the role of novel biomarkers of 21st century in patients of DM as predictor of diabetic risks or to detect earlier diabetic complications so as to aid in better understanding of disease course propose safe and effective therapeutic interventions. Various biomarkers are shown in ►Fig. 1.
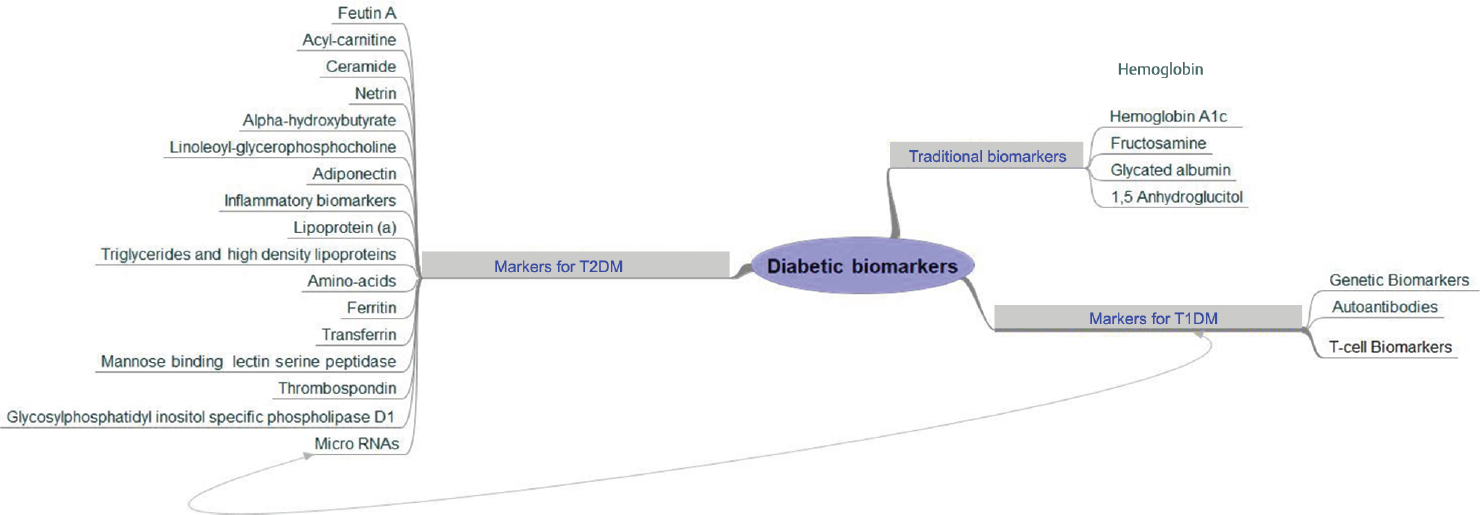
- Biomarkers in diabetes mellitus.
Traditional Biomarkers
Hemoglobin A1c
Chronic glycemia is better estimated by hemoglobin A1c (HbA1c) than glucose levels at a single time point and currently is the most commonly used biomarker for diagnosing prediabetes and diabetes. American Diabetes Association criteria for diabetes are HbA1c ≥6.5% (48 mmol/mol) and 5.7 to 6.4% (39–46 mmol/mol) for prediabetes.14 In the Norfolk prospective study, higher HbA1c levels were associated with increased CVD and all-cause mortality.15 HbA1c may be a better predictor of microvascular complications than fasting plasma glucose (FPG).16 Other advantages of HbA1c over FPG and oral glucose tolerance test (OGTT) are its greater convenience as fasting is not required, greater preanalytical stability, and less day-to-day perturbation during periods of stress and illness.17 Thus, HbA1c is particularly useful for lifestyle modification counselling as it reflects chronic exposure to glucose but, however, there is conflicting evidence regarding its usefulness in diagnosing diabetes in comparison to OGTT and FPG as it provides moderate sensitivity.17,18 The National Health and Nutrition Examination Survey and Screening for Impaired Glucose Tolerance studies showed HbA1c levels <5.7% (39 mmol/mol) correlate only 60 to 70% of subjects having normal glucose tolerance.19,20,21 Moreover, HbA1c threshold for prediabetes does not take ethnicity, body mass index (BMI), and age, all of which may significantly alter HbA1c levels under consideration.22,23,24,25 OGTT more strongly correlates with IR and insulin secretion than HbA1c.26 HbA1c is also not always a reliable measurement of average circulating glucose levels as changes in the production rate or circulating life span of red blood cells will affect HbA1c levels; for example, it may be falsely elevated in iron deficiency anemia, folate and vitamin B12 deficiency, severe hypertriglyceridemia conditions where reduced production leads to a greater percent of older cells, whereas false low values may been seen in conditions of rapid turnover of red blood cells like in HbA1c occurs in splenomegaly and end-stage renal disease.27 Hemoglobin variants, such as HbS, HbC, HbD, and HbE, may also result in overestimation or underestimation of HbA1c.28 So, HbA1c alone cannot be considered adequate for diagnosing prediabetes, and more accurate diagnosis may require confirmation with other biomarkers.29
Fructosamine
Fructosamine (FA) is a ketoamine formed by glycosylation of fructose to total serum protein, mostly albumin.30 It reflects average blood glucose concentrations over the previous 1 to 4 weeks, and thus can be a useful clinical marker of short-term glycemic fluctuation and glucose control.31 Currently, it is used as an alternate glycemic marker for diabetes screening. FA could be a valuable complementary marker in clinical conditions, where HbA1c may be inaccurate or it may also play a role in identifying fluctuating glucose levels in DM patients with stable HbA1c along with other measures.32,33 FA measurement is cost-effective and convenient as it does not require fasting34,35 and is advantageous in conditions that affect hemoglobin levels.36 However, limitations include, higher variations within the subjects37 and falsely low values are seen physiologically in young children who have lower serum protein concentration than adults and pathologically in conditions with rapid albumin turnover as in nephrotic syndrome, severe liver disease, or protein-losing enteropathy.38 Though it is useful to diagnose prediabetes, some studies also doubt its usefulness for prediabetes screening.31,39,40,41 Thus, FA can serve as a useful alternate biomarker under specific conditions.
Glycated Albumin
Glycated albumin (GA) measures the ratio of GA to total albumin and thus it is better than FA in clinical conditions that result in protein loss such as nephrotic syndrome, liver, and thyroid disease.42 It is also considered to be a better index of glycemic control than HbA1c in patients with renal failure, hemolytic anemia, and those receiving blood transfusions.43,44 Due to shorter half-life of albumin, GA reflects a shorter glycemic control, of 2 to 3 weeks as compared with HbA1c.45 The combination of GA with HbA1c is a more sensitive predictor of prediabetes than HbA1c alone.46 The speed of glycation of GA is 10 times faster than HbA1c; thus, its value better reflects variations in blood glucose and postprandial hyperglycemia in combination with HbA1c.47 GA estimation is of limited use in conditions of abnormal albumin metabolism.48 Other disadvantage is that sometimes GA may be artificially low in individuals having increased BMI, body fat mass, and high visceral fat.49
1,5-Anhydroglucitol (1,5-AG)
It is the naturally occurring, 1-deoxy form of glucose whose plasma levels are inversely correlated with plasma glucose levels. Renal proximal tubules have relatively greater affinity for glucose than 1,5-AG. Thus, plasma concentrations of 1,5-AG decrease due to its increased urinary concentration, as studied in healthy control, prediabetes, and diabetes groups.50 It has been suggested as a prediabetes marker and reflects glucose levels within the preceding 10 to 14 days. AG has the advantage of being stable, reproducible, and less costly when compared with other glycemic diagnostic tests and may be useful for identifying postprandial glycemic excursions and individuals at risk of microvascular and macrovascular complications in diabetes.51 However, plasma 1,5-AG levels can change based on diet, sex,52,53 and race and also fluctuating levels have been seen in patients receiving SGLT 2 inhibitors or those on renal replacement therapy.54,55 Also, some studies disagree with the use of 1,5-AG as a prediabetes screening tool. On the contrary, a few studies disagree with the use of 1,5-AG as a prediabetes screening tool.53,54
Novel Biomarkers for Type 2 Diabetes Mellitus
Fetuin A
Fetuin-A, also called alpha 2-Heremans Schmid glycoprotein, is a physiological inhibitor of insulin receptor tyrosine kinase and thus associated with IR, metabolic syndrome and an increased risk for T2DM.56,57 Pal et al showed that fetuin-A binds to toll-like receptor 4 (TLR4)-inflammatory signaling pathway, which results in production of inflammatory cytokines, and, thus, promotes lipid-induced IR through this interaction.58 Fetuin-A knockout mice have also demonstrated increased basal and insulin-stimulated phosphorylation of insulin receptor, increased glucose clearance, and improved insulin sensitivity.59,60 As measured by the index of homeostasis model (HOMA-IR), fetuin-A is found to be an independent determinant in the development of IR.61,63,63 It is well known that IR has been identified as the major pathophysiologic determinant of T2DM.64,65 In cross-sectional studies, genetic analyses revealed that SNPs in the fetuin-A gene (located at a susceptibility locus for T2DM) are linked to T2DM.66,67 Both the Health, Aging and Body Composition Study (Health ABC) and European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study indicate that participants with high fetuin-A levels have an increased risk of incident diabetes.68 However, there have been conflicting reports regarding association between fetuin-A concentration and CVD.69,71,72,72 Thus, taken together, fetuin-A acts as an endogenous ligand for TLR4 through which lipids induce IR and may serve as a novel therapeutic target for IR.
Acyl-Carnitine
L-carnitine, a small water-soluble molecule, plays an essential role in intermediary metabolism, by transporting long-chain fatty acids from the cytosol into the mitochondria, where their degradation takes place via β-oxidation.73 Beyond this, other crucial functions in the body are modification of acyl-(conenzyme A) CoA/CoA ratio, energy storage in the form of acetyl carnitine, anti-inflammatory and antioxidant properties. It also improves insulin sensitivity, dyslipidemia, and membrane stability.74 Recently, serum levels of acylcarnitines have been shown to be elevated in prediabetes.75,76 Lipid oversupply results in accumulation of incompletely metabolized fatty acids in the mitochondria causing “mitochondrial stress,” leading to IR.77 According to another theory, the long-chain acyl-CoAs are precursors of ceramide that is already being explored as affector of insulin resistance.78 Recent studies suggest an alternative mechanism in which fatty acid oxidation (FAO) rate outpaces that of the tricarboxylic acid cycle, resulting in the accumulation of intermediary metabolites, such as acylcarnitines, which may affect insulin sensitivity.74 A recent Canadian study reveals that an elevation in circulating medium chain acylcarnitines is associated with gestational DM and early stages of T2D onset and that this elevation directly impairs β-cell function.79 Another study also showed acylcarnitine profile, mainly including short- and long-chain acylcarnitines, was significantly associated with higher T2D risk in participants at high cardiovascular risk.80
Ceramides
Ceramides, a type of sphingolipid with long chain fatty acid of varying length, constitute major component of the biological membrane. Ceramides are available in the human system by de novo pathway, salvage pathway, and by sphingomyelin hydrolysis. Their accumulation may increase in tissues due to excessive supply of fatty acids, mostly as a result of sphingolipid salvage pathway activity.81 Apart from its structural role in biological membranes and signal transduction, ceramides can also antagonize insulin signaling by inhibiting transmission of signals through phosphatidylinositol-3 kinase and blocking activation of the anabolic enzyme Akt/PKB. They also stimulate caspase, protein kinase C, serine/threonine protein phosphatase, and cathepsin D activity.82 Thus, ceramides interfere with glucose uptake, impair storage of nutrients such as glycogen or triglyceride (Tg), activate proinflammatory cytokines, disrupt lipid metabolism, and even enhance cell death (proapoptotic). Plasma ceramides have been found to be increased in obesity and insulin resistance (IR).83 Insulin also elicits an anabolic effect on sphingolipid metabolism, resulting in increased ceramide accumulation in skeletal muscles.84 This shows a deleterious consequence of the hyperinsulinemia that accompanies IR leading to a vicious cycle. A study suggests that ceramide stearic to palmitic acid ratio Cer (d18:1/18:0)/Cer(d18:1/16:0) is an independent predictive biomarker for new onset diabetes mellitus even years before diagnosis; thus it may be modulated by lifestyle intervention.85 Since ceramides are detectable in easily accessible body fluids, they have recently been proposed as promising biomarker candidates in several diseases such as cancer, multiple sclerosis, Alzheimer's disease, and coronary artery disease and T2DM.86
Netrin
Netrin is a family of extracellular, laminin-related proteins,87 comprising of netrin-1, netrin-3, and netrin-4, and two glycosylphosphatidylinositol (GPI) anchored membrane peptides (netrin G1 and G2).88 They are expressed in the central nervous system and also in nonneural tissues such as vascular endothelial cells, pancreas, liver, spleen, lung, intestine, and kidney.89 Keeping in view its pathogenic role, netrin is emerging as a novel diagnostic and prognostic biomarker for variety of life-threatening diseases like cancers, cardiovascular diseases, acute kidney injury, and subarachnoid hemorrhage.90 As netrin-1 is involved in pancreatic morphogenesis, islet-cell migration, and rejuvenation, it was likely that they have a role in diabetes as well. A recent clinical study found a significant increase of serum Netrin-1 level in subjects with impaired fasting glucose (IFG) or T2DM compared with the control group suggesting that Netrin-1 may be used as a biomarker for their early detection.91 On the contrary, another study found that the level of Netrin-1 in diabetic patients was significantly reduced than that of healthy controls.92 However, netrins, having role in angiogenesis and inflammatory process, show a promising role as marker for early detection and prognosis of diabetic microvascular complications like retinopathy and nephropathy.90 Netrin-1 regulates corneal epithelial wound healing, inflammation response, and nerve fiber regeneration in diabetic mice, indicating the potential application for the therapy of diabetic keratopathy.91 They have also shown beneficial efficacy in the treatment of diabetic nephropathy.92
α-Hydroxybutyrate and Linoleoylglycerophosphocholine
Recently increased levels of plasma α-hydroxybutyrate (α-HB), an organic acid, and decreased levels of plasma linoleoylglycerophosphocholine (L-GPC), a lipid, have been described as joint markers of peripheral IR and glucose intolerance, as measured by the euglycemic hyperinsulinemic clamp technique—in selected subjects in the Relationship between Insulin Sensitivity and Cardiovascular Disease study, a cohort of well-phenotyped nondiabetic individuals.93 α-Hydroxybutyrate (α-HB) is a catabolic by-product of threonine, methionine, and glutathione anabolism (cysteine formation) in hepatic tissue.93 Increased oxidative stress and lipid oxidation lead to chronic shifts in glutathione synthesis resulting in elevated α-HB levels in individuals of IR.94,95 Thus, it is suggested that α-HB, a proximate product of disordered metabolism, might serve as both a predictive biomarker and prodromal sign of incipient T2DM.96
L-GPC is formed by hepatic phospholipase A2 and circulatory lecithincholesterol acyltransferase. Choline-containing phospholipids and sphingomyelins have been associated with increased risk of T2DM.97,98 L-GPC is an independent correlate of insulin sensitivity and a putative lipid-signaling molecule.93
Both the above biomarkers of insulin sensitivity are independently associated with glucose intolerance.93 However, L-GPC is a negative predictor of T2DM progression in contrast to αHB, a positive predictor.93,99 But both the biomarkers can be used in predictive models to identify subjects with impaired glucose tolerance that is a high-risk state for the development of T2DM, without performing an OGTT.97
Adiponectin
Adiponectin is a protein exclusively expressed in differentiated adipocytes, cardiac tissue, bone, mammary and salivary glands.100 It has insulin sensitizing, anti-inflammatory, and anti-atherogenic functions and it is shown to be independent predictor of diabetes.101 It also has antiangiogenic and possibly anticancer properties.102 Adiponectin may be an important modulator of insulin sensitivity through increased oxidation of fatty acid, glucose uptake and utilization in skeletal muscle and adipose tissue.103 High level of adiponectin protects glucose metabolism impairment in obese patients and decreases the risk of T2DM development.104 Level of adiponectin is found to be lower in T2DM and obese subjects.105 In a cohort-based study of subjects with impaired glucose tolerance, low adiponectin level was found to be a strong independent predictor of diabetes.106 In offspring of diabetic parents, the baseline adiponectin levels are inversely related to the risk of prediabetes and it is independent of sex or ethnicity.107 Adiponectin administration has found lower levels of plasma glucose and increased insulin sensitivity.108 On the other hand, treatment of patients with insulin increased the level of circulating adiponectin and improved insulin sensitivity.104
Inflammatory Biomarkers
Inflammatory biomarkers are involved in the activation of innate and adaptive immune systems that are recognized as key mediators in diabetes development. Specifically, acute phase proteins (C-reactive protein [CRP] or high-sensitivity [hs-CRP] and fibrinogen) and pro-inflammatory cytokines (interleukin-6 [IL-6] and tumor necrosis factor-α) are associated with increased risk of T2D.109 Elevated levels of IL-6 and CRP are useful in identifying individuals at higher risk of developing T2DM.110 Genetic variants in the innate immune system and inflammatory cascade also affect CRP and predisposition to T2DM.111,112 A meta-analysis of genome-wide association study on hs-CRP showed shared genes and pathways that are associated with IR and T2D.113,114 Tissue plasminogen activator-1 change is an independent predictor of incidence of diabetes.115 IL-18 levels also increased with progression from prediabetes to diabetes in the Gutenberg study.116 The IL-1 receptor antagonist (IL-1RA), produced by adipocytes, is an anti-inflammatory marker elevated in prediabetes and diabetes, possibly as a reactive response to inflammation. The Whitehall Study, has also shown an increase in IL-1RA in prediabetes in parallel with decreasing insulin sensitivity, increasing β-cell function, and 2-hour glucose levels, all of which occurred altogether years before the development of T2DM.116 Patients with T2DM frequently have dysfunctional coagulation profile, especially increased fibrinogen levels that are independently associated with HbA1c values.117 Fibrinogen levels were also seen to be associated with diabetic nephropathy, but not with other microvascular complications like retinopathy. With respect to macrovascular disease, the association between high plasma fibrinogen and peripheral vascular disease is seen in T1DM.118
Lipoprotein(a)
Lipoprotein (a) [Lp (a)] is a plasma lipoprotein that consists of LDL-like particle, consisting of apo (a) that is covalently attached to one molecule of apoB100 via a disulfide bond and is synthesized in liver.119 Lp(a) levels are primarily under genetic control, with genetic variants and the number of Kringle IV-2 repeats in the LPA gene accounting for much of the variability in Lp (a) concentrations.120 Elevated levels of LP(a) are proved to be independent risk factor for the development of CVD.121 Increase in Lp (a) levels may promote atherosclerosis via Lp (a)-derived cholesterol entrapment in the intima, via inflammatory cell recruitment, and/or via the binding of proinflammatory-oxidized phospholipids.122 Inverse relationship between serum Lp (a) and T2DM, prediabetes, IR, and with hyperinsulinemia has also been noted.123 Though the mechanisms through which Lp(a) might be inversely associated with diabetes risk is not yet clear, there is some evidence that Lp(a) is a marker of, or might be involved in, the development of IR.
Amino Acids
Amino acids have emerged as novel biomarkers in the identification of people at risk of T2D before overt symptoms. With advancement in technologies involving metabolic profiling, circulating amino acids may illustrate new pathways in diabetes pathophysiology like bidirectional modulation of insulin action due to crosstalk between hormonal and nutritional signals and may suggest novel mechanism by amino acids.124,125 More recent studies have demonstrated a correlation between amino acids and prediabetes, IR, and obesity.93 It is shown that serum branched chain amino acids and aromatic amino acids are significantly and positively associated with T2D.126 In addition, glutamine, methionine, cysteine, and 2aminoadipic acid are increased in insulin resistant states.127,99,129 By contrast, glycine levels are decreased in individuals with prediabetes.130,99,132 Thus, changes in these circulating amino acid levels may prove to be significant predictive markers for T2D.
Triglycerides and High-Density Lipoprotein
Elevated serum Tg and subclasses of high-density lipoprotein cholesterol (HDLC) maybe involved in the pathogenesis of diabetes. Hypertriglyceridemia has been associated with β-cell dysfunction and reduced insulin secretion in prediabetes.121 Tg levels can cause lipotoxicity within pancreatic β-cells and may lead to β-cell apoptosis by stimulating the production of ceramide and nitric oxide.121,133 Levels of small HDL-3 particles have been found to be elevated in prediabetic subjects as compared with HDLC levels. The proportion of small HDL-3 particles is positively associated with Tg and negatively associated with HDLC.134 However, unlike Tg, low HDLC concentrations may also lead to progression to diabetes from prediabetes, but its association with β-cell dysfunction is unclear.121
Ferritin and Transferrin
Both ferritin and transferrin levels are associated with hyperinsulinemia and hyperglycemia.135 A recent study in healthy women showed that higher iron stores as reflected by ferritin concentrations and the ratio of transferrin receptors to ferritin were associated with an increased risk of T2DM, independently of known diabetes risk factors.136 Iron contributes to IR through the production of highly active radical formation, damage to DNA and cell membrane integrity, β-cell oxidative stress resulting in decreased insulin secretory capacity, and interference with glucose uptake in skeletal muscles and adipocytes. Dietary iron restriction prevents the development of diabetes and loss of β-cell function.137 Not only iron stores but also iron transport is involved in the development of the metabolic syndrome and very likely of IR.138 Transferrin has been shown to be a major determinant of lipolytic activity in adipocytes by a pro-oxidative mechanism.139 and adipose tissue lipolysis has been recognized as a major determinant of IR.140 Higher levels of transferrin are usually negatively correlated with ferritin.135
Mannose-Binding Lectin Serine Peptidase
Mannan-binding lectin serine protease 1 also known as mannose-associated serine protease 1 (MASP-1) is involved in the lectin pathway of the complement system and is responsible for cleaving C4 and C2 into fragments to form a C3-convertase.141 MASP-1 is also able to cleave fibrinogen and factor XIII and may be involved in coagulation.142 Thus, elevated levels of these proteins may play a role in the enhanced thrombotic environment and consequent vascular complications in diabetes.143 It has been observed that the onset of prediabetes and IR occurred earlier in those with increased MASP-1 plasma levels. Elevated FPG and 2-hour glucose levels also have positive association with higher levels of MASP-1.144 Thus, these proteins have a potential role as a biomarker to detect early macrovascular complications in diabetes.
Thrombospondin
Thrombospondins (TSP 1–5) are a group of multifunctional secretory glycoproteins. TSP-1, a potent antiangiogenic and proatherogenic protein, has been implicated in the development of several vascular diabetic complications.145 TSP-4 is an extracellular matrix protein of the vessel wall.146 Endogenous TSP-1 protects the myocardium from infarction-induced and pressure overload-induced cardiac remodeling.147 TSP-1 is also an adipokine that is highly expressed in obese, insulin-resistant subjects; and it is highly correlated with adipose inflammation.148 In the diabetic setting, decrease in TSP increases matrix metalloproteinase (MMP-2 and MMP-9) activities and suppresses fibroblast function thus inhibiting collagen synthesis.149 They could be a novel marker for atherosclerotic burden, especially in the major subgroup of patients with concomitant diabetes.150 TSP-1 plays an important role in the development of complications in patients with diabetic nephropathy and its serum level is related to renal injury and vascular disease.151
Glycosylphosphatidylinositol Specific Phospholipase D1
Glycosylphosphatidylinositol-specific phospholipase D (GPLD1, also called GPI-PLD) is a 110- to 120-kDa N-glycosylated amphiphilic protein abundant in mammalian serum, where it associates with HDL.152 The association with HDL raises the possibility that GPI-PLD may be involved in lipid/lipoprotein metabolism. GPLD1 may be regulated by several different hormones or metabolites involving circulating insulin levels, hyperglycemia, oxidative stress, and inflammation.152,153 In addition, GPI-PLD appears to colocalize with insulin in the secretory granule,154 raising the possibility that diabetes may be associated with changes in islet production of GPI-PLD. Recent evidence suggests that GPLD1 levels are increased in circulation following the onset of diabetes induction and IR both in rats and humans, respectively, suggesting its association with diabetes-induced impairments.155,156 GPLD1 may play an important role in inflammation and in the pathogenesis of diabetes. Recent study suggests the potential role of GPLD1 as a candidate plasma protein that can be used effectively to distinguish between early stage latent autoimmune diabetes in adults and T2DM.157
Micro-RNAs
Micro-RNAs (miRNAs) are small (~22 nucleotides) noncoding RNA sequences that inhibit gene expression of specific mRNA targets.158 It has been reported that distinct miRNA expression profiles regulate various physiological and pathological conditions.159 Recently, miRs have been studied in prediabetes and found to be strongly correlated.160,161 Moreover, they could be differentially expressed in various types of DM, suggesting their potential to serve as diabetic indicators. It has been reported that miR-20b, miR-21, miR-24, miR-15a, miR-126, miR-191, miR-197, miR-223, miR-320, and miR-486 were downregulated, while miR-28–3p was upregulated in plasma of T2DM patients in comparison with non-DM individuals.162 More recent studies also detected significantly increased levels of circulating miR-146a163 and miR-126164 in newly diagnosed T2DM patients compared with healthy controls. Other miRNAs significantly elevated in T2DM and found to negatively regulate insulin expression, production, or secretion include miR9, miR29a, miR30d, miR34a, miR124a2, miR146a, and miR375.165
In patients with recent-onset T1DM, the most consistently upregulated miRNAs were miR-152, miR-181a, and miR-27b, while miR-375 was consistently downregulated in independent cohorts.166 miR-25, miR-24–3p, let-7 g-5p, and miR-93–5p were either upregulated or downregulated in various recent-onset T1DM cohorts.167
Recent studies suggest brain as a key player in glucose regulation and the pathogenesis of metabolic disorders such as T2D.168 There is evidence suggesting that increased activation of hypothalamus-pituitary-adrenal axis and sympathetic nervous system, and consequent elevation of stress hormones, may be important for the etiology and development of T2D. The association between these neuroendocrine stress response-related circulating miRNAs and T2D has been explored by Liang et al, which may act as potential biomarkers for prediabetes and IR in adults.169
Thus, our understanding of the importance of miRNAs in the pathogenesis of diabetes has grown substantially; there is increasing evidence for a potential role of miRNAs as clinical biomarkers of the T1DM, pre-DM, T2DM, and gestational DM. Moreover, discovery of new set of miRNA biomarkers might help to guide diagnostic and therapeutic decisions.
Possible Biomarkers for Type 1 Diabetes Mellitus
Immune biomarkers of T1DM are diverse. Though some of them like autoantibodies are well established, they are not discriminative enough to deal with the heterogeneity that is inherent in T1DM. As an alternative, various possible biomarkers have recently been identified that not only provide a better understanding and progression of T1DM but also yield therapeutic efficacy of immune interventions. ►Fig. 2 highlights various biomarkers in T1DM.
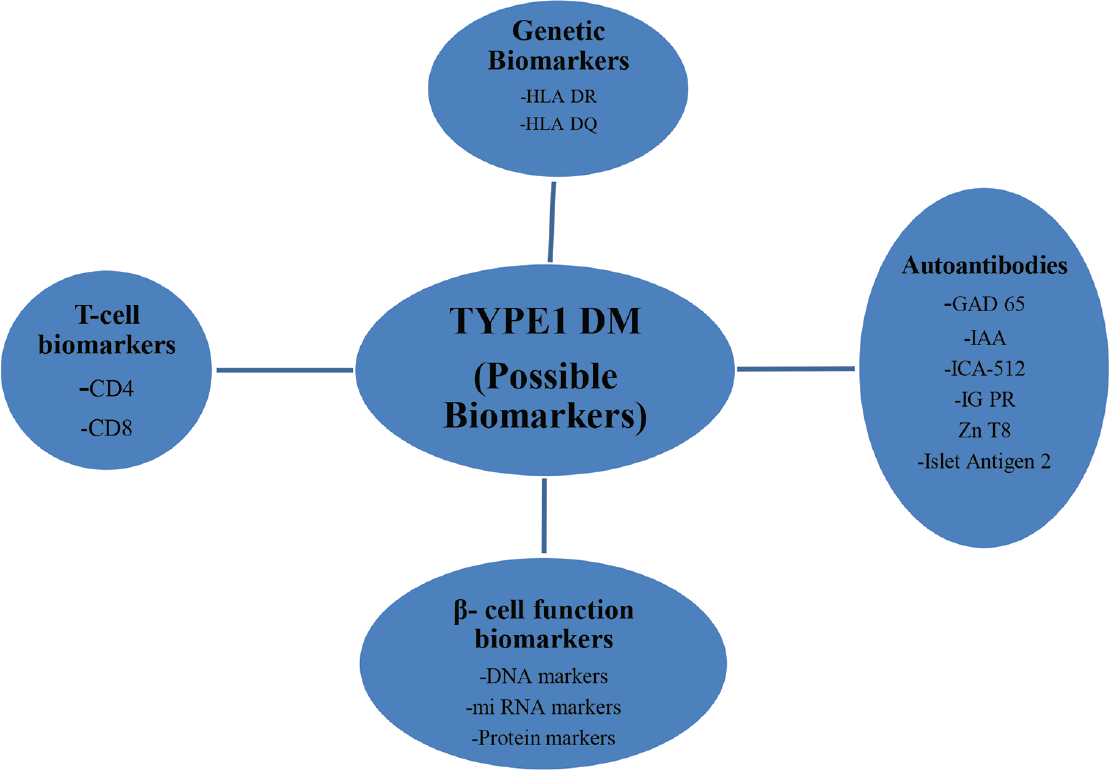
- Possible biomarkers in T1DM. GAD-65, glutamic acid decarboxylase-65; IAA, insulin-associated autoantibodies; ICA-512, internal carotid artery-512; IGPR, islet specific glucose-6-phosphatase catalytic subunit related protein; T1DM, type-1 diabetes mellitus; ZnT8, zinc transporter 8.
Genetic Biomarkers
Genetic markers may be helpful in assessing the predisposition for T1DM, where identification of over 50 loci contributing to T1DM risk has been identified in studies of large cohorts.170 For routine screening purpose, HLA typing is done. Though HLA typing alone is useful for the enrichment of future cases of T1DM, it is insufficiently sensitive and specific to be a biomarker for future prevention strategies. However, HLA-DR (DR3/4) and HLA-DQ (DQ8) genotypes are useful in predicting the risk of developing β-cell autoimmunity. The highest risk of HLADR/-DQ genotypes is present in around 30 to 40% of individuals with T1DM and around 2 to 3% of the background population.171
Autoantibodies
Autoantibodies are now widely accepted as the hallmark of T1DM. Autoantibodies against β-cell proteins and peptides for diagnosis of T1DM, are routinely used.172 The most commonly measured are autoantibodies to glutamic acid decarboxylase, insulin-associated autoantibodies, insulinoma-associated protein 2 (IA-2, previously known as ICA-512), islet specific glucose-6-phosphatase catalytic subunit related protein, a tyrosine phosphatase like protein (islet antigen-2 [IA-2] and the most recently described zinc transporter 8.173,174 However, the development of autoantibodies against multiple β-cell antigens is recognized as a critical step in the disease pathogenesis and is associated with a significantly higher T1DM risk than the presence of just a single autoantibody.175,176 Autoantibody characteristics that allow for stratification of diabetes risk include age at seroconversion, antibody number, titer, affinity, antigen specificity, and epitope binding.174,177 Recently, novel autoantibodies have been described, including those targeting neoantigens generated in β-cells under conditions of stress (e.g., immune stress and metabolic stress). These include antibodies against modified β-cell-derived peptides or proteins generated through stress-induced post-translational modifications, like citrullination.178
T-Cell Biomarkers
Current evidence suggests that T-cells are the main mediators in the pathogenesis of T1DM.179,180 Thus, T-cell biomarkers are becoming an important component of immunotherapy trials in T1DM, identifying logical targets for intervention, providing novel insights into why (or in whom) treatments succeed or fail, and providing potential for participant stratification.181
CD4 T-cells provide help, cytokines, and regulation, while CD8 T-cells produce inflammatory cytokines to drive the destructive immune response forward and kill β-cells.183 Furthermore, frequencies of islet antigen-reactive CD4 and CD8 T-cells are higher in T1DM patients compared with healthy subjects, though variable over time, and may have the potential to function as a prognostic marker for disease onset or treatment response.183,184
The role of T cells as essential cellular constituents of disease progression has motivated research consortium efforts to develop T-cell biomarkers in T1DM, with attention to two broad classes of markers, namely, (1) antigen specific (i.e., captured by assays that measure the number and/or function of T cells specific for B cell autoantigens) and (2) antigen agnostic (i.e., involving assays that measure T cell attributes without accounting for the specificity conferred by the T cell receptor).185
Conclusion and Future Perspectives
There is a vital need for the identification of more sensitive and precise biomarkers for subset of individuals with different underlying pathogenesis and difference in speed of disease progression at its earliest that will help to facilitate personalized prediction, prevention, and treatment of diabetes mellitus. Biomarkers are needed to guide our understanding of the disease process whether it is destruction of β-cells by the immune system as in T1DM or reduction in β-cell function as in T2DM. Combining biomarkers in a clinical setting may provide better sensitivity and specificity in predicting and preventing the disease. However, long-term prospective studies are needed to further to establish the utility of these biomarkers in establishing early diagnosis and management of the disease and thereby preventing complications. Furthermore, genetic studies, assessing mutations, will also provide additional insight into its association with metabolic dysregulation.186 Different omics platforms—genomics, metabolomics, proteomics, and microbiomics—and RNA sequencing-based studies, coupled with novel data science methods involving bioinformatics, data mining, imaging, machine learning, neural networks, are now revolutionizing biomarker development.187,99,189 Adoption of such methods in accordance with data protection and ethical guidelines will improve quality of life for the patients and enable better health care.
Conflict of Interest
None declared.
References
- Diabetes epidemiology as a tool to trigger diabetes research and care. Diabetologia. 1999;42(05):499-518.
- [CrossRef] [PubMed] [Google Scholar]
- Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract. 2014;103(02):137-149.
- [CrossRef] [PubMed] [Google Scholar]
- An update on the pharmacotherapy options for pediatric diabetes. Expert Opin Biol Ther. 2014;14(03):355-364.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnosis and classification of diabetes mellitus. Diabetes Care. 2007;30(Suppl. 01):S42-S47.
- [CrossRef] [PubMed] [Google Scholar]
- Implications of rising prediabetes prevalence. Diabetes Care. 2013;36(08):2139-2141.
- [CrossRef] [PubMed] [Google Scholar]
- National Diabetes Statistics Report: Estimates of Diabetes and its Burden in the United States. 2014
- [Google Scholar]
- Improving the health status of US working adults with type 2 diabetes mellitus. Dis Manag Health Outcomes. 2003;11(08):489-498.
- [CrossRef] [Google Scholar]
- Early detection and treatment of type 2 diabetes reduce cardiovascular morbidity and mortality: a simulation of the results of the Anglo-Danish Dutch study of intensive treatment in people with screen-detected diabetes in primary care (ADDITION-Europe) Diabetes Care. 2015;38(08):1449-1455.
- [CrossRef] [PubMed] [Google Scholar]
- Pathogenesis of NIDDM. A balanced overview. Diabetes Care. 1992;15(03):318-368.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular disease risk in type 2 diabetes mellitus: insights from mechanistic studies. Lancet. 2008;371(9626):1800-1809.
- [CrossRef] [PubMed] [Google Scholar]
- Circulating microRNAs as novel biomarkers for diabetes mellitus. Nat Rev Endocrinol. 2013;9(09):513-521.
- [CrossRef] [PubMed] [Google Scholar]
- Thirty-one novel biomarkers as predictors for clinically incident diabetes. PLoS One. 2010;5(04):e10100.
- [CrossRef] [PubMed] [Google Scholar]
- Standards of medical care in diabetes–2014. Diabetes Care. 2014;37(Suppl. 01):S14-S80.
- [CrossRef] [PubMed] [Google Scholar]
- No evidence of an increased mortality risk associated with low levels of glycated haemoglobin in a non-diabetic UK population. Diabetologia. 2011;54(08):2025-2032.
- [CrossRef] [PubMed] [Google Scholar]
- Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 1997;20(07):1183-1197.
- [CrossRef] [PubMed] [Google Scholar]
- The pros and cons of diagnosing diabetes with A1C. Diabetes Care. 2011;34(Suppl. 02):S184-S190.
- [CrossRef] [PubMed] [Google Scholar]
- Screening for diabetes and pre-diabetes with proposed A1C-based diagnostic criteria. Diabetes Care. 2010;33(10):2184-2189.
- [CrossRef] [PubMed] [Google Scholar]
- Implications of alternative definitions of prediabetes for prevalence in U.S. adults. Diabetes Care. 2011;34(02):387-391.
- [CrossRef] [PubMed] [Google Scholar]
- Use of HbA1c for diagnoses of diabetes and prediabetes: comparison with diagnoses based on fasting and 2-hr glucose values and effects of gender, race, and age. Metab Syndr Relat Disord. 2014;12(05):258-268.
- [CrossRef] [PubMed] [Google Scholar]
- Implications of using hemoglobin A1C for diagnosing diabetes mellitus. Am J Med. 2011;124(05):395-401.
- [CrossRef] [PubMed] [Google Scholar]
- Moving to an A1C-based diagnosis of diabetes has a different impact on prevalence in different ethnic groups. Diabetes Care. 2010;33(03):580-582.
- [CrossRef] [PubMed] [Google Scholar]
- Diabetes Prevention Program Research Group. Differences in A1C by race and ethnicity among patients with impaired glucose tolerance in the Diabetes Prevention Program. Diabetes Care. 2007;30(10):2453-2457.
- [CrossRef] [PubMed] [Google Scholar]
- Increased hemoglobin A1c threshold for prediabetes remarkably improving the agreement between A1c and oral glucose tolerance test criteria in obese population. J Clin Endocrinol Metab. 2015;100(05):1997-2005.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of aging on A1C levels in individuals without diabetes: evidence from the Framingham Offspring Study and the National Health and Nutrition Examination Survey 2001-2004. Diabetes Care. 2008;31(10):1991-1996.
- [CrossRef] [PubMed] [Google Scholar]
- A1C between 5.7 and 6.4% as a marker for identifying pre-diabetes, insulin sensitivity and secretion, and cardiovascular risk factors: the Insulin Resistance Atherosclerosis Study (IRAS). Diabetes Care. 2010;33(09):2104-2109.
- [CrossRef] [PubMed] [Google Scholar]
- Pitfalls in hemoglobin A1c measurement: when results may be misleading. J Gen Intern Med. 2014;29(02):388-394.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of hemoglobin variants and chemically modified derivatives on assays for glycohemoglobin. Clin Chem. 2001;47(02):153-163.
- [CrossRef] [PubMed] [Google Scholar]
- Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Clin Chem. 2011;57(06):e1-e47.
- [CrossRef] [PubMed] [Google Scholar]
- Plasma protein glycation as measured by fructosamine assay. Clin Chem. 1987;33(07):1141-1146.
- [CrossRef] [PubMed] [Google Scholar]
- Fructosamine is a useful indicator of hyperglycaemia and glucose control in clinical and epidemiological studies–cross-sectional and longitudinal experience from the AMORIS cohort. PLoS One. 2014;9(10):e111463.
- [CrossRef] [PubMed] [Google Scholar]
- Serum fructosamine as an index of glycaemia: comparison with glycated haemoglobin in diabetic and non-diabetic individuals. Pract Diabetes Int. 1987;4:126-128.
- [CrossRef] [Google Scholar]
- Can fructosamine be a surrogate for HbA(1c) in evaluating the achievement of therapeutic goals in diabetes? Diabetes Metab. 2001;27(5 Pt 1):598-603.
- [Google Scholar]
- Usefulness of fructosamine for monitoring outpatients with diabetes. Am J Med Sci. 1999;318(05):316-323.
- [CrossRef] [PubMed] [Google Scholar]
- The value of serum fructosamine in comparison with oral glucose tolerance test (OGTT) as a screening test for detection of gestational diabetes mellitus. J Obstet Gynaecol. 2000;20(02):136-138.
- [CrossRef] [PubMed] [Google Scholar]
- Serum glycated albumin to guide the diagnosis of diabetes mellitus. PLoS One. 2016;11(01):e0146780.
- [CrossRef] [PubMed] [Google Scholar]
- Glycated albumin to glycated hemoglobin ratio is a sensitive indicator of blood glucose variability in patients with fulminant type 1 diabetes. Intern Med. 2012;51(11):1315-1321.
- [CrossRef] [PubMed] [Google Scholar]
- Classification of variant forms of haemoglobin according to the ratio of glycated haemoglobin to glycated albumin. Ann Clin Biochem. 2012;49(Pt 5):441-444.
- [CrossRef] [PubMed] [Google Scholar]
- Alternative biomarkers for assessing glycemic control in diabetes: fructosamine, glycated albumin, and 1,5- anhydroglucitol. Ann Pediatr Endocrinol Metab. 2015;20(02):74-78.
- [CrossRef] [PubMed] [Google Scholar]
- Fructosamine is a useful indicator of hyperglycaemia and glucose control in clinical and epidemiological studies–cross-sectional and longitudinal experience from the AMORIS cohort. PLoS One. 2014;9(10):e111463.
- [Google Scholar]
- Rational diagnoses of diabetes: the comparison of 1,5-anhydroglucitol with other glycemic markers. Springerplus. 2015;4:587.
- [CrossRef] [PubMed] [Google Scholar]
- Advantages and pitfalls of fructosamine and glycated albumin in the diagnosis and treatment of diabetes. J Diabetes Sci Technol. 2015;9(02):169-176.
- [CrossRef] [PubMed] [Google Scholar]
- HbA1c, fructosamine, and glycated albumin in the detection of dysglycaemic conditions. Curr Diabetes Rev. 2016;12(01):14-19.
- [CrossRef] [PubMed] [Google Scholar]
- Nontraditional markers of glycemia: associations with microvascular conditions. Diabetes Care. 2011;34(04):960-967.
- [CrossRef] [PubMed] [Google Scholar]
- Usefulness of glycated albumin as an indicator of glycemic control status in patients with hemolytic anemia. Clin Chim Acta. 2011;412(03/04):253-257.
- [CrossRef] [PubMed] [Google Scholar]
- A1C combined with glycated albumin improves detection of prediabetes in Africans: the Africans in America Study. Diabetes Care. 2016;39(02):271-277.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of glycemic variability in well-controlled type 2 diabetes mellitus. Diabetes Technol Ther. 2013;15(06):455-460.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of glycated hemoglobin and fetal hemoglobin-adjusted HbA1c measurements in infants. Pediatr Diabetes. 2013;14(04):267-272.
- [CrossRef] [PubMed] [Google Scholar]
- Utility of glycated albumin for the diagnosis of diabetes mellitus in a Japanese population study: results from the Kyushu and Okinawa Population Study (KOPS) Diabetologia. 2011;54(12):3028-3036.
- [CrossRef] [PubMed] [Google Scholar]
- Serum 1,5-anhydroglucitol (GlycoMark): a short-term glycemic marker. Diabetes Technol Ther. 2003;5(03):355-363.
- [CrossRef] [PubMed] [Google Scholar]
- Serum 1,5-anhydroglucitol (1,5 AG): new clinical marker for glycemic control. Diabetes Res Clin Pract. 1994;24(Suppl):S261-S268.
- [CrossRef] [PubMed] [Google Scholar]
- Serum 1,5-anhydroglucitol level as a screening tool for diabetes mellitus in a community- based population at high risk of diabetes. Acta Diabetol. 2017;54(05):425-431.
- [CrossRef] [PubMed] [Google Scholar]
- 1,5-Anhydroglucitol in diabetes mellitus. Endocrine. 2013;43(01):33-40.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of canagliflozin, a sodium-glucose cotransporter 2 inhibitor, on measurement of serum 1,5-anhydroglucitol. J Diabetes. 2014;6(04):378-380.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of renal function on serum concentration of 1,5-anhydroglucitol in type 2 diabetic patients in chronic kidney disease stages I-III: a comparative study with HbA1c and glycated albumin. J Diabetes. 2016;8(05):712-719.
- [CrossRef] [PubMed] [Google Scholar]
- Serum fetuin-A levels in patients with type 2 diabetes mellitus. Ann UMCS Sect DDD. 2010;2(14):93-99.
- [Google Scholar]
- Association of low fetuin-A (AHSG) concentrations in serum with cardiovascular mortality in patients on dialysis: a cross-sectional study. Lancet. 2003;361(9360):827-833.
- [CrossRef] [PubMed] [Google Scholar]
- Fetuin-A acts as an endogenous ligand of TLR4 to promote lipid-induced insulin resistance. Nat Med. 2012;18(08):1279-1285.
- [CrossRef] [PubMed] [Google Scholar]
- α2-Heremans-Schmid glycoprotein/fetuin-A is associated with insulin resistance and fat accumulation in the liver in humans. Diabetes Care. 2006;29(04):853-857.
- [CrossRef] [PubMed] [Google Scholar]
- Fetuin-A and its relation to metabolic syndrome and fatty liver disease in obese children before and after weight loss. J Clin Endocrinol Metab. 2008;93(11):4479-4485.
- [CrossRef] [PubMed] [Google Scholar]
- Serum fetuin-A is correlated with metabolic syndrome in middle-aged and elderly Chinese. Atherosclerosis. 2011;216(01):180-186.
- [CrossRef] [PubMed] [Google Scholar]
- Serum fetuin-A in metabolic and inflammatory pathways in patients with myocardial infarction. Eur J Clin Invest. 2011;41(07):703-709.
- [CrossRef] [PubMed] [Google Scholar]
- Fetuin-A levels in obesity: differences in relation to metabolic syndrome and correlation with clinical and laboratory variables. Arch Med Sci. 2012;8(05):826-833.
- [CrossRef] [PubMed] [Google Scholar]
- Extended-release niacin decreases serum fetuin-A concentrations in individuals with metabolic syndrome. Diabetes Metab Res Rev. 2009;25(05):427-434.
- [CrossRef] [PubMed] [Google Scholar]
- Role of fetuin-A in atherosclerosis associated with diabetic patients. J Pharm Pharmacol. 2012;64(12):1703-1708.
- [CrossRef] [PubMed] [Google Scholar]
- AHSG tag single nucleotide polymorphisms associate with type 2 diabetes and dyslipidemia: studies of metabolic traits in 7,683 white Danish subjects. Diabetes. 2008;57(05):1427-1432.
- [CrossRef] [PubMed] [Google Scholar]
- A synonymous coding polymorphism in theα2-Heremans-schmid glycoprotein gene is associated with type 2 diabetes in French Caucasians. Diabetes. 2005;54(08):2477-2481.
- [CrossRef] [PubMed] [Google Scholar]
- Challenges in the management of type 2 diabetes mellitus and cardiovascular risk factors in obese subjects: what is the evidence and what are the myths? Int J Endocrinol. 2013;2013:856793.
- [CrossRef] [PubMed] [Google Scholar]
- Serum fetuin-A in nondialyzed patients with diabetic nephropathy: relationship with coronary artery calcification. Kidney Int. 2005;67(03):1070-1077.
- [CrossRef] [PubMed] [Google Scholar]
- Serum fetuin-A levels are associated with the presence and severity of coronary artery disease in patients with type 2 diabetes. Biomarkers. 2013;18(02):160-164.
- [CrossRef] [PubMed] [Google Scholar]
- Decreased serum fetuin-A levels are associated with coronary artery diseases. Intern Med. 2010;49(13):1281-1285.
- [CrossRef] [PubMed] [Google Scholar]
- Fetuin-A is inversely associated with coronary artery calcification in community-living persons: the Multi-Ethnic Study of Atherosclerosis. Clin Chem. 2012;58(05):887-895.
- [CrossRef] [PubMed] [Google Scholar]
- Carnitine transport and fatty acid oxidation. Biochim Biophys Acta. 2016;1863(10):2422-2435.
- [CrossRef] [PubMed] [Google Scholar]
- Role of carnitine and its derivatives in the development and management of type 2 diabetes. Nutr Diabetes. 2018;8(01):8.
- [CrossRef] [PubMed] [Google Scholar]
- Human serum acylcarnitine profiles in different glucose tolerance states. Diabetes Res Clin Pract. 2014;104(03):376-382.
- [CrossRef] [PubMed] [Google Scholar]
- Serum levels of acylcarnitines are altered in prediabetic conditions. PLoS One. 2013;8(12):e82459.
- [CrossRef] [PubMed] [Google Scholar]
- Lipid oversupply, selective insulin resistance, and lipotoxicity: molecular mechanisms. Biochim Biophys Acta. 2010;1801(03):252-265.
- [CrossRef] [PubMed] [Google Scholar]
- Acyl-CoA synthesis, lipid metabolism and lipotoxicity. Biochim Biophys Acta. 2010;1801(03):246-251.
- [CrossRef] [PubMed] [Google Scholar]
- Elevated medium-chain acylcarnitines are associated with gestational diabetes mellitus and early progression to type 2 diabetes and induce pancreaticβ-cell dysfunction. Diabetes. 2018;67(05):885-897.
- [CrossRef] [PubMed] [Google Scholar]
- Plasma acylcarnitines and risk of type 2 diabetes in a Mediterranean population at high cardiovascular risk. J Clin Endocrinol Metab. 2019;104(05):1508-1519.
- [CrossRef] [PubMed] [Google Scholar]
- Ceramides as modulators of cellular and whole-body metabolism. J Clin Invest. 2011;121(11):4222-4230.
- [CrossRef] [PubMed] [Google Scholar]
- Phosphatidylinositol 3-kinase/AKT pathway regulates the endoplasmic reticulum to Golgi traffic of ceramide in glioma cells: a link between lipid signaling pathways involved in the control of cell survival. J Biol Chem. 2009;284(08):5088-5096.
- [CrossRef] [PubMed] [Google Scholar]
- The role of ceramides in insulin resistance. Front Endocrinol (Lausanne). 2019;10:577.
- [CrossRef] [PubMed] [Google Scholar]
- Insulin increases ceramide synthesis in skeletal muscle. J Diabetes Res. 2014;2014:765784.
- [CrossRef] [PubMed] [Google Scholar]
- Ceramide stearic to palmitic acid ratio predicts incident diabetes. Diabetologia. 2018;61(06):1424-1434.
- [CrossRef] [PubMed] [Google Scholar]
- Ceramides as novel disease biomarkers. Trends Mol Med. 2019;25(01):20-32.
- [CrossRef] [PubMed] [Google Scholar]
- UNC5B receptor deletion exacerbates tissue injury in response to AKI. J Am Soc Nephrol. 2014;25(02):239-249.
- [CrossRef] [PubMed] [Google Scholar]
- Role of Netrin-1 signaling in nerve regeneration. Int J Mol Sci. 2017;18(03):491.
- [CrossRef] [PubMed] [Google Scholar]
- Netrin as a novel biomarker and its therapeutic implications in diabetes mellitus and diabetes-associated complications. J Diabetes Res. 2018;2018:8250521.
- [CrossRef] [PubMed] [Google Scholar]
- Netrin-1 promotes diabetic corneal wound healing through molecular mechanisms mediated via the adenosine 2B receptor. Sci Rep. 2018;8(01):5994.
- [CrossRef] [PubMed] [Google Scholar]
- Protective role for netrin-1 during diabetic nephropathy. J Mol Med (Berl). 2013;91(09):1071-1080.
- [CrossRef] [PubMed] [Google Scholar]
- RISC Study Group. alpha-hydroxybutyrate is an early biomarker of insulin resistance and glucose intolerance in a nondiabetic population. PLoS One. 2010;5(05):e10883.
- [CrossRef] [PubMed] [Google Scholar]
- The formation of 2-hydroxybutyric acid in experimental animals. Clin Chim Acta. 1975;58(01):23-32.
- [CrossRef] [PubMed] [Google Scholar]
- Reduction of alpha-ketobutyrate by human serum. Nature. 1960;188:1110-1111.
- [CrossRef] [PubMed] [Google Scholar]
- Early metabolic markers of the development of dysglycemia and type 2 diabetes and their physiological significance. Diabetes. 2013;62(05):1730-1737.
- [CrossRef] [PubMed] [Google Scholar]
- α-Hydroxybutyric acid is a selective metabolite biomarker of impaired glucose tolerance. Diabetes Care. 2016;39(06):988-995.
- [CrossRef] [PubMed] [Google Scholar]
- Product inhibition of secreted phospholipase A2 may explain lysophosphatidylcholines' unexpected therapeutic properties. J Inflamm (Lond). 2008;5:17.
- [CrossRef] [PubMed] [Google Scholar]
- A novel fasting blood test for insulin resistance and prediabetes. J Diabetes Sci Technol. 2013;7(01):100-110.
- [CrossRef] [PubMed] [Google Scholar]
- Adiponectin, obesity, and cancer: clash of the bigwigs in health and disease. Int J Mol Sci. 2019;20(10):2519.
- [CrossRef] [PubMed] [Google Scholar]
- Adiponectin trajectories before type 2 diabetes diagnosis: Whitehall II study. Diabetes Care. 2012;35(12):2540-2547.
- [CrossRef] [PubMed] [Google Scholar]
- Adiponectin-induced antiangiogenesis and antitumor activity involve caspase-mediated endothelial cell apoptosis. Proc Natl Acad Sci U S A. 2004;101(08):2476-2481.
- [CrossRef] [PubMed] [Google Scholar]
- Adiponectin, leptin, and fatty acids in the maintenance of metabolic homeostasis through adipose tissue crosstalk. Cell Metab. 2016;23(05):770-784.
- [CrossRef] [PubMed] [Google Scholar]
- Adiponectin, a therapeutic target for obesity, diabetes, and endothelial dysfunction. Int J Mol Sci. 2017;18(06):1321.
- [CrossRef] [PubMed] [Google Scholar]
- Hypoadiponectinemia in obese subjects with type II diabetes: a close association with central obesity indices. J Res Med Sci. 2011;16(06):713-723.
- [Google Scholar]
- Plasma adiponectin is an independent predictor of type 2 diabetes in Asian Indians. Diabetes Care. 2003;26(12):3226-3229.
- [CrossRef] [PubMed] [Google Scholar]
- Adiponectin levels predict prediabetes risk: the Pathobiology of Prediabetes in A Biracial Cohort (POP-ABC) study. BMJ Open Diabetes Res Care. 2016;4(01):e000194.
- [CrossRef] [PubMed] [Google Scholar]
- Adiponectin: action, regulation and association to insulin sensitivity. Obes Rev. 2005;6(01):13-21.
- [CrossRef] [PubMed] [Google Scholar]
- Use of readily accessible inflammatory markers to predict diabetic kidney disease. Front Endocrinol (Lausanne). 2018;9:225.
- [CrossRef] [PubMed] [Google Scholar]
- Inflammatory markers and risk of type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2013;36(01):166-175.
- [CrossRef] [PubMed] [Google Scholar]
- Genetic polymorphisms of innate immunity-related inflammatory pathways and their association with factors related to type 2 diabetes. BMC Med Genet. 2011;12:95.
- [CrossRef] [PubMed] [Google Scholar]
- Genetic variation, C-reactive protein levels, and incidence of diabetes. Diabetes. 2007;56(03):872-878.
- [CrossRef] [PubMed] [Google Scholar]
- LifeLines Cohort Study,. CHARGE Inflammation Working Group. Genome analyses of >200,000 individuals identify 58 loci for chronic inflammation and highlight pathways that link Inflammation and complex disorders. Am J Hum Genet. 2018;103(05):691-706.
- [Google Scholar]
- Common variants of inflammatory cytokine genes are associated with risk of nephropathy in type 2 diabetes among Asian Indians. PLoS One. 2009;4(04):e5168.
- [CrossRef] [PubMed] [Google Scholar]
- The Insulin Resistance Atherosclerosis Study (IRAS). Relative contribution of insulin and its precursors to fibrinogen and PAI-1 in a large population with different states of glucose tolerance. Arterioscler Thromb Vasc Biol. 1999;19(03):562-568.
- [CrossRef] [PubMed] [Google Scholar]
- Profile of the immune and inflammatory response in individuals with prediabetes and type 2 diabetes. Diabetes Care. 2015;38(07):1356-1364.
- [CrossRef] [PubMed] [Google Scholar]
- A study of plasma fibrinogen level in type-2 diabetes mellitus and its relation to glycemic control. Indian J Hematol Blood Transfus. 2012;28(02):105-108.
- [CrossRef] [PubMed] [Google Scholar]
- DCCT/ECIC STUDY GROUP. Fibrinogen is a marker for nephropathy and peripheral vascular disease in type 1 diabetes: studies of plasma fibrinogen and fibrinogen gene polymorphism in the DCCT/EDIC cohort. Diabetes Care. 2003;26(05):1439-1448.
- [CrossRef] [PubMed] [Google Scholar]
- A new serum type system in man—the LP system. Acta Pathol Microbiol Scand. 1963;59:369-382.
- [CrossRef] [PubMed] [Google Scholar]
- Human genetics and the causal role of lipoprotein(a) for various diseases. Cardiovasc Drugs Ther. 2016;30(01):87-100.
- [CrossRef] [PubMed] [Google Scholar]
- Increased levels of triglycerides, BMI and blood pressure and low physical activity increase the risk of diabetes in Swedish women. A prospective 18-year follow-up of the BEDA study. Diabet Med. 2004;21(06):615-622.
- [CrossRef] [PubMed] [Google Scholar]
- European Atherosclerosis Society Consensus Panel. Lipoprotein(a) as a cardiovascular risk factor: current status. Eur Heart J. 2010;31(23):2844-2853.
- [CrossRef] [PubMed] [Google Scholar]
- Serum lipoprotein (a) concentrations are inversely associated with T2D, prediabetes, and insulin resistance in a middle-aged and elderly Chinese population. J Lipid Res. 2015;56(04):920-926.
- [CrossRef] [PubMed] [Google Scholar]
- Bidirectional modulation of insulin action by amino acids. J Clin Invest. 1998;101(07):1519-1529.
- [CrossRef] [PubMed] [Google Scholar]
- An amino acid profile to predict diabetes.? Nat Med. 2011;17(04):418-420.
- [CrossRef] [PubMed] [Google Scholar]
- Branched-chain and aromatic amino acid profiles and diabetes risk in Chinese populations. Sci Rep. 2016;6:20594.
- [CrossRef] [PubMed] [Google Scholar]
- 2-Aminoadipic acid is a biomarker for diabetes risk. J Clin Invest. 2013;123(10):4309-4317.
- [CrossRef] [PubMed] [Google Scholar]
- Metabolite profiles and the risk of developing diabetes. Nat Med. 2011;17(04):448-453.
- [CrossRef] [PubMed] [Google Scholar]
- Lifestyle and cardiovascular disease risk factors as determinants of total cysteine in plasma: the Hordaland Homocysteine Study. Am J Clin Nutr. 1999;70(06):1016-1024.
- [CrossRef] [PubMed] [Google Scholar]
- Novel biomarkers for pre-diabetes identified by metabolomics. Mol Syst Biol. 2012;8:615.
- [CrossRef] [PubMed] [Google Scholar]
- Metabolomics in prediabetes and diabetes: a systematic review and metaanalysis. Diabetes Care. 2016;39(05):833-846.
- [CrossRef] [PubMed] [Google Scholar]
- Glutathione synthesis is diminished in patients with uncontrolled diabetes and restored by dietary supplementation with cysteine and glycine. Diabetes Care. 2011;34(01):162-167.
- [CrossRef] [PubMed] [Google Scholar]
- Serum triglyceride levels correlated with the rate of change in insulin secretion over two years in prediabetic subjects. Ann Nutr Metab. 2014;64(01):38-43.
- [CrossRef] [PubMed] [Google Scholar]
- Small high-density lipoprotein (HDL) subclasses are increased with decreased activity of HDL-associated phospholipase A2 in subjects with prediabetes. Lipids. 2013;48(06):547-555.
- [CrossRef] [PubMed] [Google Scholar]
- Insulin Resistance Syndrome (DESIR) Study Group. Ferritin and transferrin are both predictive of the onset of hyperglycemia in men and women over 3 years: the data from an epidemiological study on the Insulin Resistance Syndrome (DESIR) study. Diabetes Care. 2006;29(09):2090-2094.
- [CrossRef] [PubMed] [Google Scholar]
- Body iron stores in relation to risk of type 2 diabetes in apparently healthy women. JAMA. 2004;291(06):711-717.
- [CrossRef] [PubMed] [Google Scholar]
- Dietary iron restriction or iron chelation protects from diabetes and loss of beta-cell function in the obese (ob/ob lep-/-) mouse. Am J Physiol Endocrinol Metab. 2010;298(06):E1236-E1243.
- [CrossRef] [PubMed] [Google Scholar]
- DESIR Study Group. Ferritin and transferrin are associated with metabolic syndrome abnormalities and their change over time in a general population: Data from an Epidemiological Study on the Insulin Resistance Syndrome (DESIR) Diabetes Care. 2007;30(07):1795-1801.
- [CrossRef] [PubMed] [Google Scholar]
- Transferrin and iron contribute to the lipolytic effect of serum in isolated adipocytes. Diabetes. 2004;53(10):2535-2541.
- [CrossRef] [PubMed] [Google Scholar]
- Insulin resistance in type 2 diabetes: role of fatty acids. Diabetes Metab Res Rev. 2002;18(Suppl. 02):S5-S9.
- [CrossRef] [PubMed] [Google Scholar]
- The role of mannose-binding lectin-associated serine protease-3 in activation of the alternative complement pathway. J Immunol. 2011;187(07):3751-3758.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of MASP-1 of the complement system on activation of coagulation factors and plasma clot formation. PLoS One. 2012;7(04):e35690.
- [CrossRef] [PubMed] [Google Scholar]
- Association between mannose-binding lectin and vascular complications in type 1 diabetes. Diabetes. 2004;53(06):1570-1576.
- [CrossRef] [PubMed] [Google Scholar]
- MASP1, THBS1, GPLD1 and ApoA-IV are novel biomarkers associated with prediabetes: the KORA F4 study. Diabetologia. 2016;59(09):1882-1892.
- [CrossRef] [PubMed] [Google Scholar]
- Thrombospondin-1 as an endogenous inhibitor of angiogenesis and tumor growth. J Cell Mol Med. 2002;6(01):1-12.
- [CrossRef] [PubMed] [Google Scholar]
- Thrombospondin-4 in tissue remodeling. Matrix Biol 2019:75-76. 300-313
- [CrossRef] [PubMed] [Google Scholar]
- Endogenous thrombospondin 1 protects the pressure-overloaded myocardium by modulating fibroblast phenotype and matrix metabolism. Hypertension. 2011;58(05):902-911.
- [CrossRef] [PubMed] [Google Scholar]
- Thrombospondin-1 is an adipokine associated with obesity, adipose inflammation, and insulin resistance. Diabetes. 2008;57(02):432-439.
- [CrossRef] [PubMed] [Google Scholar]
- Thrombospondin-1: the good, the bad, and the complicated. Circ Res. 2013;113(12):1272-1274.
- [CrossRef] [PubMed] [Google Scholar]
- Thrombospondin-4 increases with the severity of peripheral arterial disease and is associated with diabetes. Heart Vessels. 2020;35(01):52-58.
- [CrossRef] [PubMed] [Google Scholar]
- Correlation of enhanced thrombospondin-1 expression, TGF-beta signalling and proteinuria in human type-2 diabetic nephropathy. Nephrol Dial Transplant. 2008;23(12):3880-3887.
- [CrossRef] [PubMed] [Google Scholar]
- Increased expression of GPI-specific phospholipase D in mouse models of type 1 diabetes. Am J Physiol Endocrinol Metab. 2001;281(01):E147-E154.
- [CrossRef] [PubMed] [Google Scholar]
- Glycosylphosphatidylinositol-specific phospholipase D is expressed by macrophages in human atherosclerosis and colocalizes with oxidation epitopes. Circulation. 1999;99(22):2876-2882.
- [CrossRef] [PubMed] [Google Scholar]
- Regulation of glycosylphosphatidylinositol-specific phospholipase D secretion from beta TC3 cells. Endocrinology. 1997;138(02):819-826.
- [CrossRef] [PubMed] [Google Scholar]
- Insulin reduces serum glycosylphosphatidylinositol phospholipase D levels in human type I diabetic patients and streptozotocin diabetic rats. Mol Genet Metab. 2002;75(02):154-161.
- [CrossRef] [PubMed] [Google Scholar]
- Insulin resistance is associated with increased serum levels of glycosylphosphatidylinositol-specific phospholipase D. Metabolism. 2004;53(02):138-139.
- [CrossRef] [PubMed] [Google Scholar]
- The clinical significance of glycoprotein phospholipase D levels in distinguishing early stage latent autoimmune diabetes in adults and type 2 diabetes. PLoS One. 2016;11(06):e0156959.
- [CrossRef] [PubMed] [Google Scholar]
- MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(02):281-297.
- [CrossRef] [PubMed] [Google Scholar]
- The diverse functions of microRNAs in animal development and disease. Dev Cell. 2006;11(04):441-450.
- [CrossRef] [PubMed] [Google Scholar]
- Peripheral blood microRNA-15a is a potential biomarker for type 2 diabetes mellitus and pre-diabetes. Mol Med Rep. 2015;12(05):7485-7490.
- [CrossRef] [PubMed] [Google Scholar]
- Serum miR-23a, a potential biomarker for diagnosis of pre-diabetes and type 2 diabetes. Acta Diabetol. 2014;51(05):823-831.
- [CrossRef] [PubMed] [Google Scholar]
- Plasma microRNA profiling reveals loss of endothelial miR-126 and other microRNAs in type 2 diabetes. Circ Res. 2010;107(06):810-817.
- [CrossRef] [PubMed] [Google Scholar]
- Increased microRNA-146a levels in plasma of patients with newly diagnosed type 2 diabetes mellitus. PLoS One. 2013;8(09):e73272.
- [CrossRef] [PubMed] [Google Scholar]
- Plasma miR-126 is a potential biomarker for early prediction of type 2 diabetes mellitus in susceptible individuals. BioMed Res Int. 2013;2013:761617.
- [CrossRef] [PubMed] [Google Scholar]
- Significance of serum microRNAs in pre-diabetes and newly diagnosed type 2 diabetes: a clinical study. Acta Diabetol. 2011;48(01):61-69.
- [CrossRef] [PubMed] [Google Scholar]
- Circulating levels of microRNA from children with newly diagnosed type 1 diabetes and healthy controls: evidence that miR-25 associates to residual beta-cell function and glycaemic control during disease progression. Exp Diabetes Res. 2012;2012:896362.
- [CrossRef] [PubMed] [Google Scholar]
- miRNA-375 a sensor of glucotoxicity is altered in the serum of children with newly diagnosed type 1 diabetes. J Diabetes Res. 2016;2016:1869082.
- [CrossRef] [PubMed] [Google Scholar]
- Is the brain a key player in glucose regulation and development of type2 diabetes? Front Physiol. 2019;10:457.
- [CrossRef] [PubMed] [Google Scholar]
- Identification of neuroendocrine stress response-related circulating microRNAs as biomarkers for type 2 diabetes mellitus and insulin resistance. Front Endocrinol (Lausanne). 2018;9:132.
- [CrossRef] [PubMed] [Google Scholar]
- Genetics of type 1 diabetes. Curr Opin Genet Dev. 2018;50:7-16.
- [CrossRef] [PubMed] [Google Scholar]
- TEDDY Study Group. Genetic scores to stratify risk of developing multiple islet autoantibodies and type 1 diabetes: a prospective study in children. PLoS Med. 2018;15(04):e1002548.
- [CrossRef] [PubMed] [Google Scholar]
- Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care. 2015;38(10):1964-1974.
- [CrossRef] [PubMed] [Google Scholar]
- The cation efflux transporter ZnT8 (Slc30A8) is a major autoantigen in human type 1 diabetes. Proc Natl Acad Sci U S A. 2007;104(43):17040-17045.
- [CrossRef] [PubMed] [Google Scholar]
- Prediction of type I diabetes in first-degree relatives using a combination of insulin, GAD, and ICA512bdc/IA-2 autoantibodies. Diabetes. 1996;45(07):926-933.
- [CrossRef] [PubMed] [Google Scholar]
- Combined analysis of autoantibodies improves prediction of IDDM in islet cell antibody-positive relatives. Diabetes. 1994;43(11):1304-1310.
- [CrossRef] [PubMed] [Google Scholar]
- Predicting type 1 diabetes using biomarkers. Diabetes Care. 2015;38(06):989-996.
- [CrossRef] [PubMed] [Google Scholar]
- Understanding type 1 diabetes through proteomics. Expert Rev Proteomics. 2017;14(07):571-580.
- [CrossRef] [PubMed] [Google Scholar]
- T cells in type 1 diabetes: instructors, regulators and effectors: a comprehensive review. J Autoimmun. 2016;66:7-16.
- [CrossRef] [PubMed] [Google Scholar]
- The case for an autoimmune aetiology of type 1 diabetes. Clin Exp Immunol. 2016;183(01):8-15.
- [CrossRef] [PubMed] [Google Scholar]
- Surrogate end points in the design of immunotherapy trials: emerging lessons from type 1 diabetes. Nat Rev Immunol. 2010;10(02):145-152.
- [CrossRef] [PubMed] [Google Scholar]
- Autoreactive CD8 T cells associated with beta cell destruction in type 1 diabetes. Proc Natl Acad Sci U S A. 2005;102(51):18425-18430.
- [CrossRef] [PubMed] [Google Scholar]
- Simultaneous detection of circulating autoreactive CD8+ T-cells specific for different islet cell-associated epitopes using combinatorial MHC multimers. Diabetes. 2010;59(07):1721-1730.
- [CrossRef] [PubMed] [Google Scholar]
- Demonstration of islet-autoreactive CD8 T cells in insulitic lesions from recent onset and long-term type 1 diabetes patients. J Exp Med. 2012;209(01):51-60.
- [CrossRef] [PubMed] [Google Scholar]
- Immunological biomarkers for the development and progression of type 1 diabetes. Diabetologia. 2018;61(11):2252-2258.
- [CrossRef] [PubMed] [Google Scholar]
- The Early Diabetes Intervention Program–is early actually late? Diabetes Metab Res Rev. 2014;30(08):654-658.
- [CrossRef] [PubMed] [Google Scholar]
- Plasma metabolomics identifies markers of impaired kidney function: a meta-analysis of 1,984 Europeans with type 2 diabetes. J Am Soc Nephrol 2018:535.
- [Google Scholar]
- Urinary proteomics and precision medicine for chronic kidney disease: current status and future perspectives. Proteomics Clin Appl. 2019;13(02):e1800176.
- [CrossRef] [PubMed] [Google Scholar]
- Application of urinary proteomics as possible risk predictor of renal and cardiovascular complications in patients with type 2-diabetes and microalbuminuria. J Diabetes Complications. 2018;32(12):1133-1140.
- [CrossRef] [PubMed] [Google Scholar]





